Replacing noble-metal catalysts for the oxygen evolution reaction (OER) with active and stable electrocatalysts made from earth-abundant elements could make alkaline water photoelectrolysis more sustainable. While there are some heteroatom-doped carbon materials for this application, such as transitional metal-N (TM–Nx) doped nanocarbon catalysts, their catalytic performance is not sufficient for practical use.
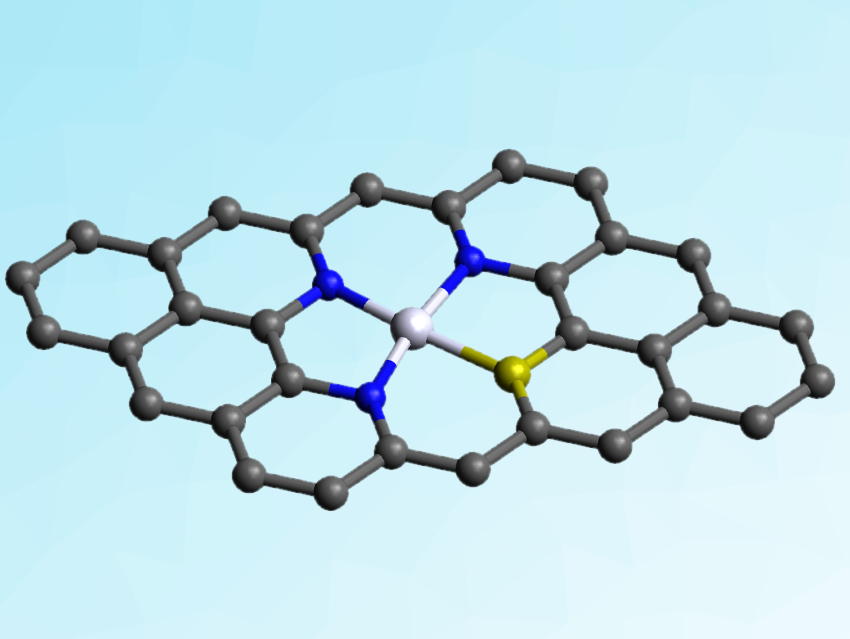
Yang Hou, Zhejiang University, Hangzhou, China, and Technische Universität Dresden, Germany, Xinliang Feng, Technische Universität Dresden, and colleagues have developed a 2D nanocarbon hybrid electrocatalyst for the OER. The catalyst consists of atomically dispersed Ni atoms coordinated with three nitrogen and one sulfur atoms in porous carbon nanosheets (S|NiNx-PC). It was synthesized from dicyandiamide, thiophene, NiCl2, and an exfoliated graphene foil.
The resulting nanosheets electrode have an outstanding electrocatalytic activity and durability for OER. They have a low overpotential of 1.51 V at 10 mA cm–2, the lowest among all heteroatom- and/or transition metal-doped carbon electrocatalysts for OER reported so far and even surpassing the state-of-the-art commercial Ir/C catalyst. This performance can be attributed to the hybrid catalyst’s porous architecture and well-distributed active sites. The incorporated sulfur atoms can effectively tune the hybridization state of the Ni and N atoms and enhance the electron transfer.
- Atomically dispersed nickel–nitrogen–sulfur species anchored on porous carbon nanosheets for efficient water oxidation,
Yang Hou, Ming Qiu, Min Gyu Kim, Pan Liu, Gyutae Nam, Tao Zhang, Xiaodong Zhuang, Bin Yang, Jaephil Cho, Mingwei Chen, Chris Yuan, Lecheng Lei, Xinliang Feng,
Nat. Commun. 2019.
https://doi.org/10.1038/s41467-019-09394-5