1,2-Cyclobutadiene, which contains an allene group in a four-membered ring, is a non-existent, unstable compound due to its very high ring strain. Synthesizing similar compounds with four-membered rings, e.g., 1-metallacyclobuta-2,3-dienes, is challenging.
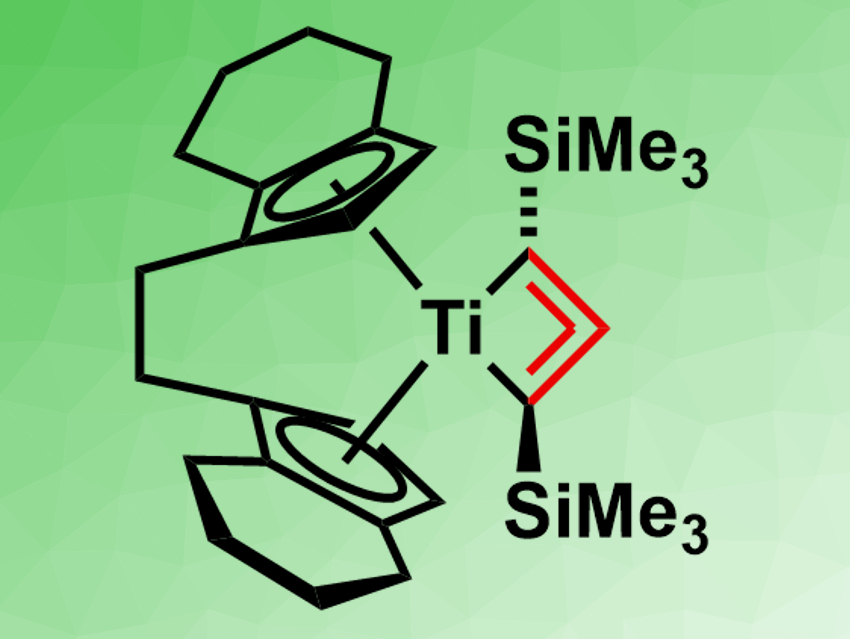
Torsten Beweries, Leibniz-Institut für Katalyse e.V., University Rostock, Germany, have prepared a 1-titanacyclobuta-2,3-diene complex. The team reacted [rac-(ebthi)TiCl2] and [Li2(Me3SiC3SiMe3)] in pentane at room temperature (rac-ebthi = rac-1,2-ethylene-1,1′-bis(η5-tetrahydroindenyl)). They obtained the complex [rac-(ebthi)Ti(Me3SiC3SiMe3)] (pictured) in moderate yields as red crystals.
The team used density functional theory (DFT) and wave function theory calculations to investigate the binding situation in the unusual complex. They found that the molecule can be described as a combination of a TiIII radical and an organic radical, with antiferromagnetic coupling between the radicals. The compound can react with ketones and aldehydes to give enynes. According to the researchers, further investigations of the complex’s reactivity will be carried out.
- 1-Titanacyclobuta-2,3-diene – an Elusive Four-membered Cyclic Allene,
Fabian Reiß, Melanie Reiß, Jonas Bresien, Anke Spannenberg, Haijun Jiao, Wolfgang Baumann, Perdita Arndt, Torsten Beweries,
Chem. Sci. 2019.
https://doi.org/10.1039/c9sc01002e