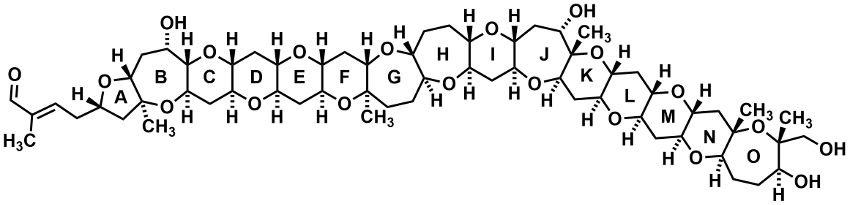
Gymnocin B (pictured) is the second-largest contiguous marine ladder polyether. It has been isolated from the dinoflagellate Karenia mikimotoi in 2005 [1]. It consists of 15 cyclic ethers and has cytotoxic properties. Until now, there has been only a single attempt at the synthesis of just the N and O rings of gymnocin B.
Satapanawat Sittihan and Timothy F. Jamison, Massachusetts Institute of Technology (MIT), Cambridge, USA, have performed the first total synthesis of gymnocin B. The team used a two-phase approach. The first phase is inspired by the proposed biosynthesis of marine ladder polyethers via epoxide-opening cascades. Four different epoxide-opening cascade reactions were used to create 10 of the cyclic ether rings. These cascade reactions were bromonium-initiated for the AB ring system, base-mediated for the C and D rings, catalyzed by a Lewis acid for the FGH ring system, and promoted by water for the K, L, and M rings.
In the second phase of the synthesis, the resulting subunits were connected by Suzuki–Miyaura coupling reactions to give the desired product. The overall synthesis has 45 steps (longest linear sequence). According to the researchers, this two-phase approach could be useful for the synthesis of other marine ladder polyethers.
- Total Synthesis of the Marine Ladder Polyether Gymnocin B,
Satapanawat Sittihan, Timothy F. Jamison,
J. Am. Chem. Soc. 2019, 141, 11239–11244.
https://doi.org/10.1021/jacs.9b04696
Reference
- [1] Gymnocin-B with the largest contiguous polyether rings from the red tide dinoflagellate, Karenia (formerly Gymnodinium) mikimotoi,
Masayuki Satake, Yoshihisa Tanaka, Yuki Ishikura, Yasukatsu Oshima, Hideo Naoki, Takeshi Yasumoto,
Tetrahedron Lett. 2005, 46, 3537–3540.
https://doi.org/10.1016/j.tetlet.2005.03.115




