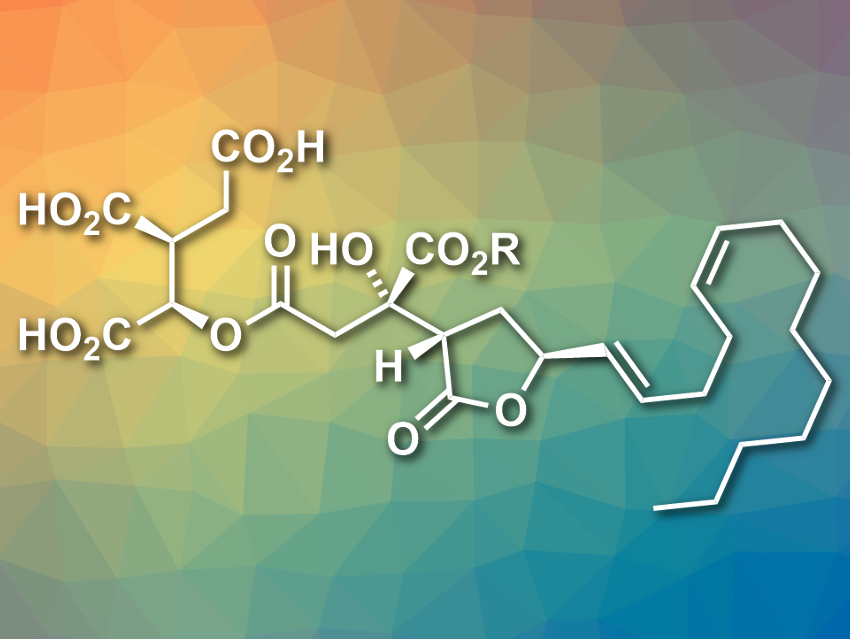
Fungi produce a class of natural products that feature a citric acid group with an alkyl substituent at the 2-position—the alkyl citrates. The citrafungins belong to this class of compounds. Citrafungin A had been synthesized and characterized, but the specific rotation of the synthesized compound did not match that of the natural product [1–3]. This points to an error in the assignment of the compound’s absolute configuration.
Mark A. Rizzacasa, The University of Melbourne, Australia, and colleagues have proposed a new, corrected structure for citrafungin A (pictured, R = H) and performed a highly stereoselective total synthesis of the compound, which confirms this structure. The team’s synthesis has twelve steps, including a formal [2 + 2]-cycloaddition, an oxa-Michael/cyclobutanone ring-opening cascade, and an asymmetric vinylzinc addition as key transformations. The team obtained 20 mg of the desired product.
The 13C NMR spectrum and the specific rotation of the synthesized product are in good agreement with those of the natural product, which confirms the structure reassignment. According to the team, their synthesis approach for complex alkyl citrates could also allow the production of higher oxidized examples of this class of natural products.
- Total Synthesis and Stereochemical Reassignment of Citrafungin A,
Zongjia Chen, Angus Robertson, Jonathan M. White, Mark A. Rizzacasa,
Org. Lett. 2019.
https://doi.org/10.1021/acs.orglett.9b03830
References
- [1] Total Synthesis of Citrafungin A,
Mumen F. A. Amer, Keisuke Takahashi, Jun Ishihara, and Susumi Hatakeyama,
Heterocycles 2007, 72, 181–185.
https://doi.org/10.3987/com-06-s(k)44 - [2] Total Synthesis of Citrafungin A,
Frederick Calo, Jeffery Richardson, Anthony G. M. Barrett,
J. Org. Chem. 2008, 73, 9692–9697.
https://doi.org/10.1021/jo801708q - [3] Formal Total Synthesis of (+)-Citrafungin A,
Sammi Tsegay, Helmut Hügel, Mark A. Rizzacasa,
Australian J. Chem. 2009, 62, 676–682.
https://doi.org/10.1071/ch09182