Transition metal dichalcogenides (TMDCs) have many useful properties and make up the largest class of 2D materials. One way to tune the properties of TMDCs for specific applications is to functionalize their surface with small molecules. However, how these molecules bind near the surface when nanopores (caused by sulfur vacancies) are present is not well understood.
Grace G. D. Han, Brandeis University, Waltham, MA, USA, and colleagues have used annular dark-field scanning transmission electron microscopy (ADF-STEM) to study how solution-deposited molecules bind to the edges and surface regions around nanopores in MoS2 monolayers. The researchers fabricated nanopores with clean edges and a controllable mean diameter using time-dependent large-area electron beam exposure.
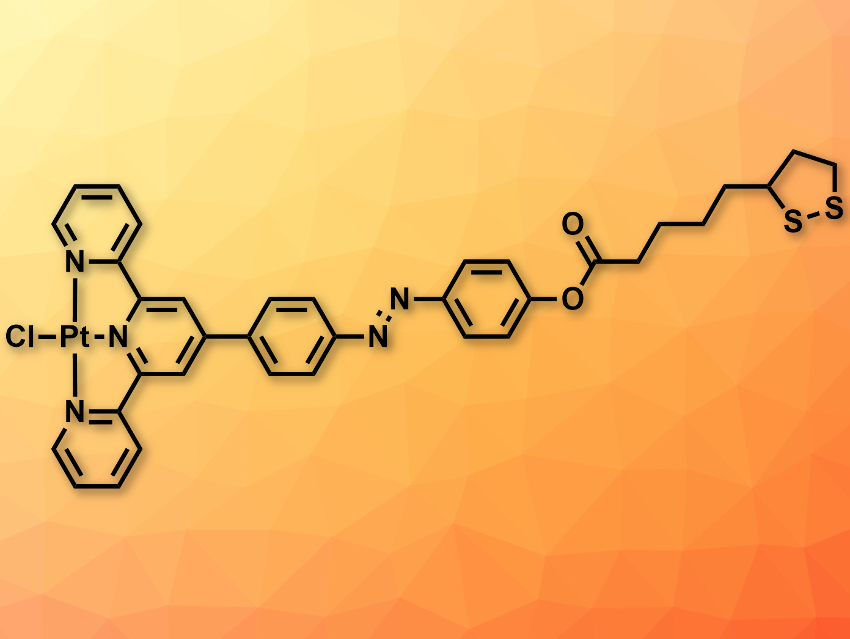
The team designed the organic attaching molecule (pictured) with a dithiolane end group so that it easily binds the Mo-terminated sites around the nanopore. The structure of the attachable organic ligand also includes a platinum marker unit at the other end of the molecule so that it can be detected by the ADF-STEM setup. The platinum atoms were then used to track the binding around the edges of the MoS2 monolayer and the molecular conformation of the molecules upon binding.
The researchers found that the molecules reside mostly on the surface of the monolayer, with one end attached to the edge of the nanopore. The preference for this orientation over dangling in the free space in the pore has been attributed to van-der-Waals interactions between the MoS2 basal plane and the aromatic core of the ligand.
- Direct Imaging of Individual Molecular Binding to Clean Nanopore Edges in 2D Monolayer MoS2,
Mihael A. Gerkman, Ja Kyung Lee, Xiang Li, Qianyang Zhang, Maurice Windley, Maria V. Fonseca, Yang Lu, Jamie H. Warner, Grace G. D. Han,
ACS Nano 2019.
https://doi.org/10.1021/acsnano.9b06061