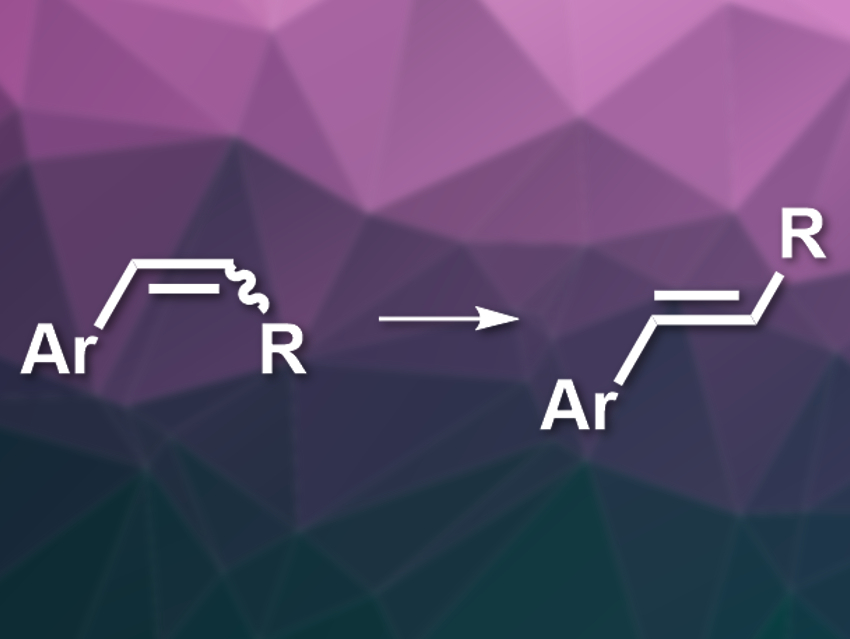
Substituted styrenes (i.e., phenylethenes) are useful building blocks in synthetic organic chemistry and the chemical industry. These compounds can be made via a variety of methods, but existing approaches often suffer from poor E/Z selectivity. The resulting E– and Z-isomers can also be difficult to separate. This means methods that can, for example, selectively isomerize Z– to E-alkenes are useful to convert E/Z mixtures into isomerically pure compounds. However, the available reactions for this type of isomerization have problems such as substrate restrictions or high catalyst loadings.
Yuanzhi Xia, Wenzhou University, China, and colleagues have developed an efficient cobalt-catalyzed Z-to-E isomerization of β-substituted styrenes. The team prepared E/Z-mixtures of a variety of β-substituted styrenes by Wittig olefinations of the corresponding aldehydes. Then they used CoCl2 as the catalyst, together with an amido-diphosphine ligand (bis(2-(diphenylphosphanyl)phenyl)amine, PNP), NaBHEt3 as a reductant, and toluene as the solvent for the isomerization (pictured).
The reaction was performed at room temperature and the desired E-styrenes were obtained from the E/Z-mixtures in good to excellent yields. The required catalyst loading is low (0.1 %) and the reaction can be performed on a gram scale.
- Cobalt-Catalyzed Z to E Isomerization of Alkenes: An Approach to (E)-β-Substituted Styrenes,
Hongmei Liu, Man Xu, Cheng Cai, Jianhui Chen, Yugui Gu, Yuanzhi Xia,
Org. Lett. 2020.
https://doi.org/10.1021/acs.orglett.0c00072