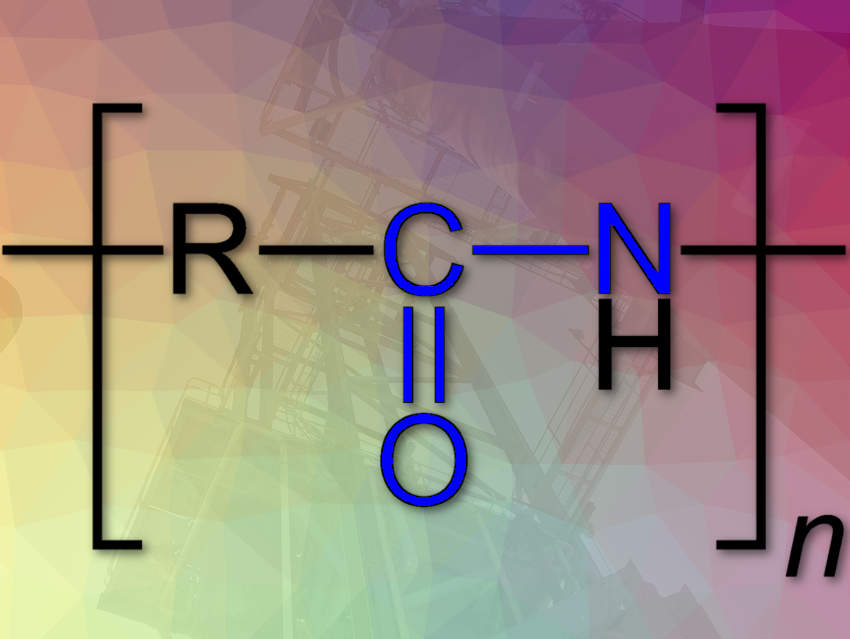
BASF closed the acquisition of Solvay’s polyamide (PA 6.6) business. Through the backward integration into the key raw material adiponitrile (ADN; (CH₂)₄(CN)₂), BASF will now be present along the entire value chain for polyamide 6.6 and improve its supply reliability. The business will be integrated into the Performance Materials and Monomers divisions of BASF.
The transaction between Solvay and BASF includes eight production sites in Germany, France, China, India, South Korea, Brazil, and Mexico, R&D centers and technical consultation centers in Asia, North and South America, and shares in two joint ventures (JV) in France: Solvay’s 50 % share in the Butachimie JV with Invista, Wichita, Kansas, USA, to produce ADN and hexamethylenediamine (HMD), and a 51 % share in the new Alsachimie JV between BASF and DOMO Chemicals, Ghent-Zwijnaarde, Belgium, to produce adipic acid. At closing, approximately 700 Solvay employees join BASF. The Alsachimie JV between BASF and DOMO Chemicals in France employs approximately 650 employees, the Butachimie JV between BASF and Invista has approximately 400 employees.
The purchase price acquired by BASF on a cash and debt-free basis amounts to €1.3 billion; sales generated in 2018 were about €1.0 billion. In September 2017, BASF signed an agreement with Solvay on the acquisition of Solvay’s global polyamide business, subject to the approval of the relevant antitrust authorities. The E.U. Commission approved the acquisition of the polyamide business, subject to certain conditions, on January 18, 2019. These conditions required the sale of parts of the original transaction scope to a third party, specifically Solvay’s production plants and innovation competencies in the engineering plastics field in Europe. DOMO Chemicals, Leuna, Germany, was approved by the E.U. Commission as the buyer. The transaction between Solvay and DOMO Chemicals also closed on January 31, 2020.
- BASF SE, Ludwigshafen, Germany
- Solvay S.A., Brussels, Belgium