Suzuki–Miyaura cross-coupling reactions are commonly used to create C–C bonds. They are catalyzed by palladium, which is rare and expensive. Researchers have tried to use alternative, cheap, earth-abundant metals for this type of reaction, with some success at high catalyst concentrations. However, for the production of, e.g., pharmaceuticals, residual metals have to be removed after the reaction—and the high catalyst loadings required for these processes make this difficult. Catalysts that work even at very low loadings, would, thus, be useful.
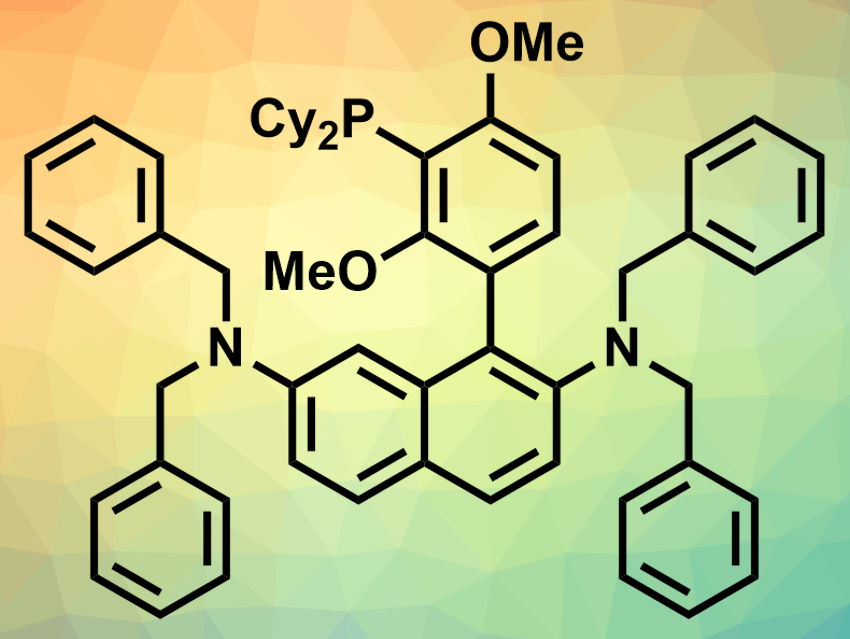
Donald H. Aue, Bruce H. Lipshutz, University of California Santa Barbara, USA, and colleagues have developed a new ligand, N2Phos (pictured), that can be used in Suzuki–Miyaura couplings with low palladium loadings (0.10–0.25 mol%). The ligand combines a biaryl skeleton with a phosphine unit and two bulky substituted amino groups. The team synthesized the ligand starting from 2,7-dibromonaphthalene, which was subjected to a double amination and a subsequent bromination. A cross-coupling reaction was then used to create the biaryl unit. Finally, a lithiation followed by a phosphine insertion gave the desired product.
The researchers used the new ligand together with Pd(OAc)2 to catalyze Suzuki–Miyaura cross-couplings between chloro-, bromo-, or iodoaryls and aryl boronic acids. They found that the developed ligand system gave good yields of the biaryl products at low catalyst loadings. The catalyst can also be used in water, which allows for environmentally friendly transformations.
- N2Phos – an easily made, highly effective ligand designed for ppm level Pd-catalyzed Suzuki–Miyaura cross couplings in water,
Nnamdi Akporji, Ruchita R. Thakore, Margery Cortes-Clerget, Joel Andersen, Evan Landstrom, Donald H. Aue, Fabrice Gallou, Bruce H. Lipshutz,
Chem. Sci. 2020.
https://doi.org/10.1039/d0sc00968g