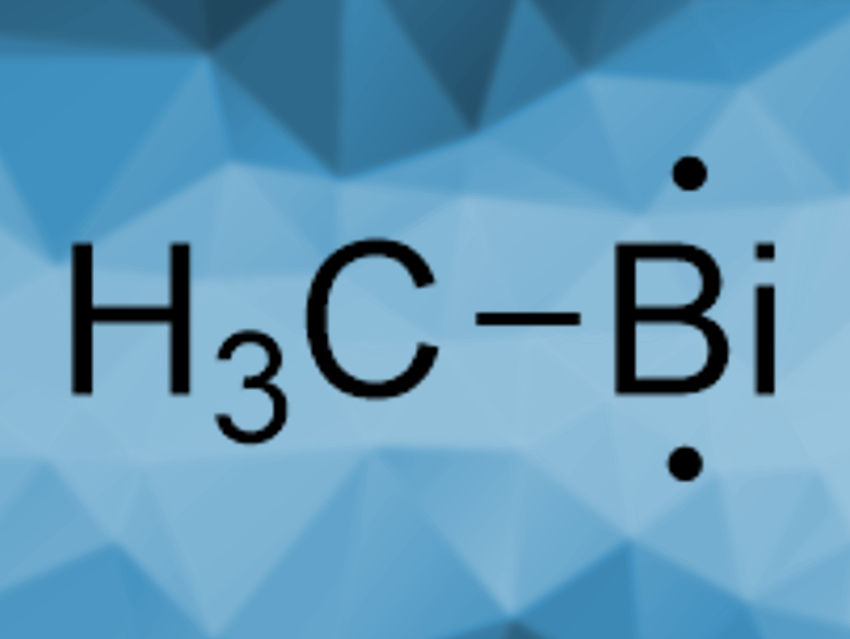Low-valent group 15 compounds, such as nitrenes or phophinidenes, are generally highly reactive, and thus, challenging to isolate. They are electron-deficient and can be stabilized by Lewis bases as ligands. The synthesis of such compounds becomes more difficult for the heavier elements in the group. Until now, all known examples of mononuclear organometallic Bi(I) compounds, so-called bismuthinidenes, have been Lewis-base adducts.
Bernd Engels, Ingo Fischer, Crispin Lichtenberg, University of Würzburg, Germany, and colleagues have synthesized and characterized the first free organometallic bismuthinidene, BiCH3 (pictured). The compound was prepared from Bi(CH3)3 in the gas phase by a thermally induced, stepwise abstraction of two of the methyl groups. BiMe3 was diluted with Ar in a microreactor and then pyrolyzed. The products were analyzed using photoelectron-photoion coincidence spectroscopy (PEPICO) and mass spectrometry (MS).
The team observed the formation of the demethylated products Bi(CH3)2· and BiCH3, as well as Bi2. The latter stems from the dimerization of “naked” Bi atoms. There are three possible structures for the free methylbismuth BiCH3: a singlet bismuthinidene, a triplet bismuthinidene, or a methylenebismuthane of the type H2C=BiH. Based on the spectral data, the team found that the ground state of BiCH3 is a triplet bismuthinidene. This stands in contrast to the Lewis-base-stabilized bismuthinidenes studied so far, which all have singlet ground states.
- Methylbismuth: an Organometallic Bismuthinidene Biradical,
Deb Pratim Mukhopadhyay, Domenik Schleier, Sara Wirsing, Jacqueline Ramler, Dustin Kaiser, Engelbert Reusch, Patrick Hemberger, Tobias Preitschopf, Ivo Krummenacher, Bernd Engels, Ingo Fischer, Crispin Lichtenberg,
Chem. Sci. 2020.
https://doi.org/10.1039/d0sc02410d