Benzene derivatives are important compounds in organic chemistry. The synthesis of benzene variants with elements other than carbon in the six-membered ring is an interesting field of research. Silicon, as carbon’s heavier analogue, is promising, but generally, synthesizing stable silicon-substituted benzenes is challenging. The use of specifically tailored, sterically demanding substituents has allowed successful syntheses of silabenzenes, 1,2-disilabenzenes, 1,4-disilabenzenes, and hexasilabenzenes.
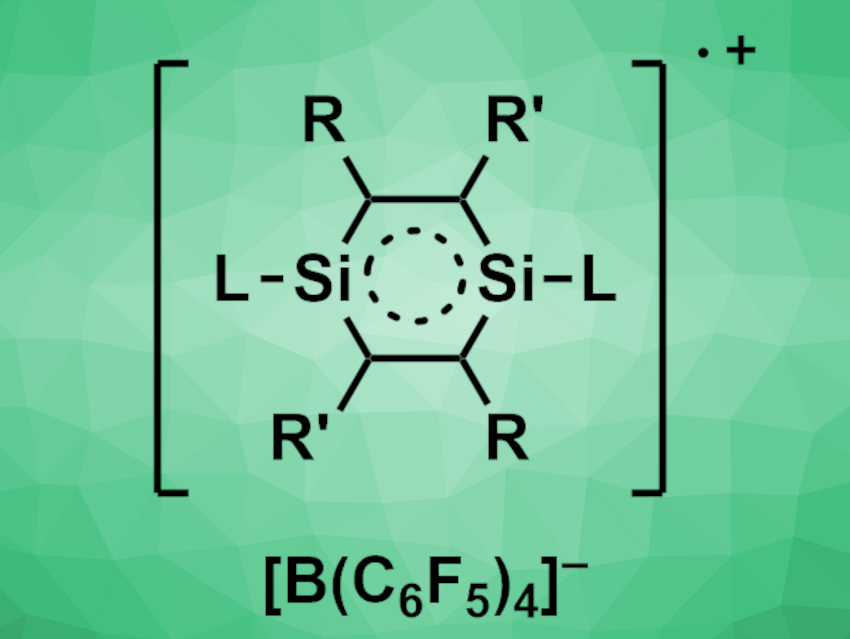
Hongping Zhu, Xiamen University, China, Herbert W. Roesky, University of Göttingen, Germany, Li Zhang, Guangxi University of Science and Technology, Liuzhou, China, Gengwen Tan, Soochow University, Suzhou, China, and colleagues have found that the 1,4-disilabenzene (Me3SiC2)2(Me3Si)2C4Si2(L)2 (L = PhC(NtBu)2) can be converted to the stable radical cation [(Me3SiC2)2(Me3Si)2C4Si2(L)2]·+[B(C6F5)4]− (pictured) or the dication [(Me3SiC2)2(Me3Si)2C4Si2(L)2]2+·2[OSO2CF3]− by the transfer of one or two electrons, respectively. The team first reacted (LSi:)2 with a functionalized alkyne to obtain the 1,4-disilabenzene.
The 1,4-disilabenzene was then reacted with [Ph3C]+[B(C6F5)4]− to give the radical cation or with AgOSO2CF3 to give the dication. All three species are sensitive to air and moisture, but stable under an inert atmosphere at room temperature. They were characterized using X-ray crystallography, UV–Vis spectroscopy, electron paramagnetic resonance (EPR) spectroscopy, and elemental analysis. The researchers found that the parent 1,4-disilabenzene has an open-shell singlet diradical character. Both the 1,4-disilabenzene and the radical cation show evidence of π-electron delocalization.
- Stable Radical Cation and Dication of a 1,4-Disilabenzene,
Yilin Chen, Jiancheng Li, Yiling Zhao, Li Zhang, Gengwen Tan, Hongping Zhu, Herbert W. Roesky,
J. Am. Chem. Soc. 2021.
https://doi.org/10.1021/jacs.0c12908