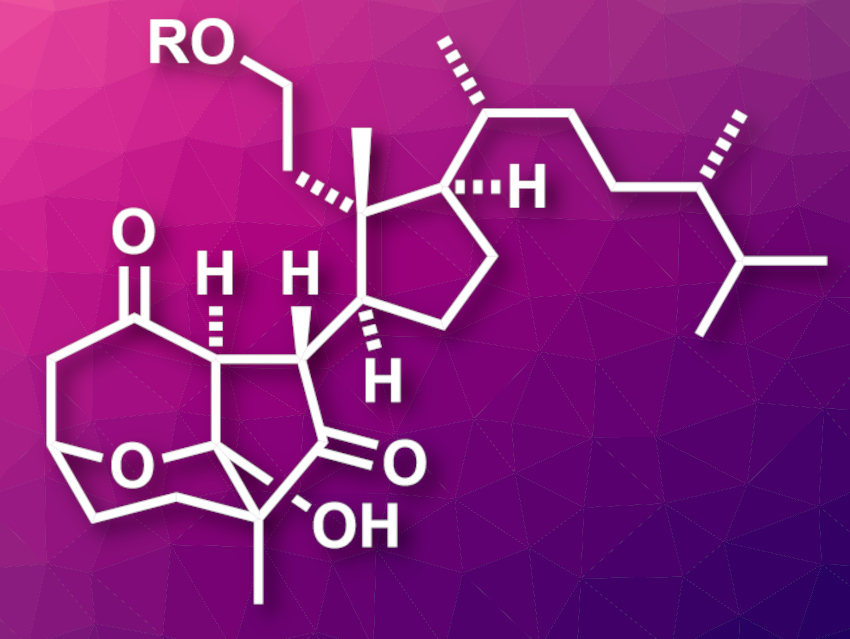
Rearranged steroids are a class of natural products with a broad structural diversity and interesting biological activities. Secosteroids are examples of this type of compound, where at least one ring in the corresponding steroid is cleaved. Pinnigorgiols B and E (pictured, R = H for pinnigorgiol B and R = Ac for pinnigorgiol E) are 9,11-secosteroids featuring a tricyclic framework with a γ-diketone moiety, which are challenging to synthesize.
Jinghan Gui, Shanghai Institute of Organic Chemistry, University of the Chinese Academy of Sciences, and colleagues have performed the first synthesis of pinnigorgiols B and E from inexpensive, commercially available ergosterol. The team’s strategy was inspired by a proposed biosynthetic pathway to pinnigorgiols B and E, in which the related compound pinnisterol E undergoes rearrangements to form the tricyclic framework of the targeted pinnigorgiols in only a few steps. They developed a similar approach that uses a semipinacol rearrangement, an oxidative ring-cleavage, and an acyl radical cyclization.
The team started from ergosterol, which was converted to dehydroergosterol, dihydroxylated, and hydrogenated. The resulting intermediate was subjected to a semipinacol rearrangement to create a 7,5-bicyclic framework. After cleaving the remaining six-ring via ozonolysis and an oxidative cleavage of the seven-ring, the team introduced a thioester, which was cleaved to create an acyl radical. Pinnigorgiol E was then obtained from this radical, presumably via an acyl radical cyclization/hemiketalization cascade that creates the tricyclic γ-diketone framework. Hydrolysis of pinnigorgiol E then gave pinnigorgiol B.
- Concise Synthesis of 9,11-Secosteroids Pinnigorgiols B and E,
Xinghui Li, Zeliang Zhang, Huafang Fan, Yinlong Miao, Hailong Tian, Yucheng Gu, Jinghan Gui,
J. Am. Chem. Soc. 2021.
https://doi.org/10.1021/jacs.0c13426