Nanographenes, or large polycyclic aromatic hydrocarbons (PAHs), such as one-dimensional graphene nanoribbons, often have interesting electronic and optical properties and could be useful, e.g., in electronic applications or photodetectors. Introducing strain into their aromatic cores can lead to structures that are twisted out of plane, which leads to higher solubilities, and thus, to systems that are easier to characterize than their planar equivalents. However, synthesizing large, twisted graphene nanoribbons using organic synthesis is challenging.
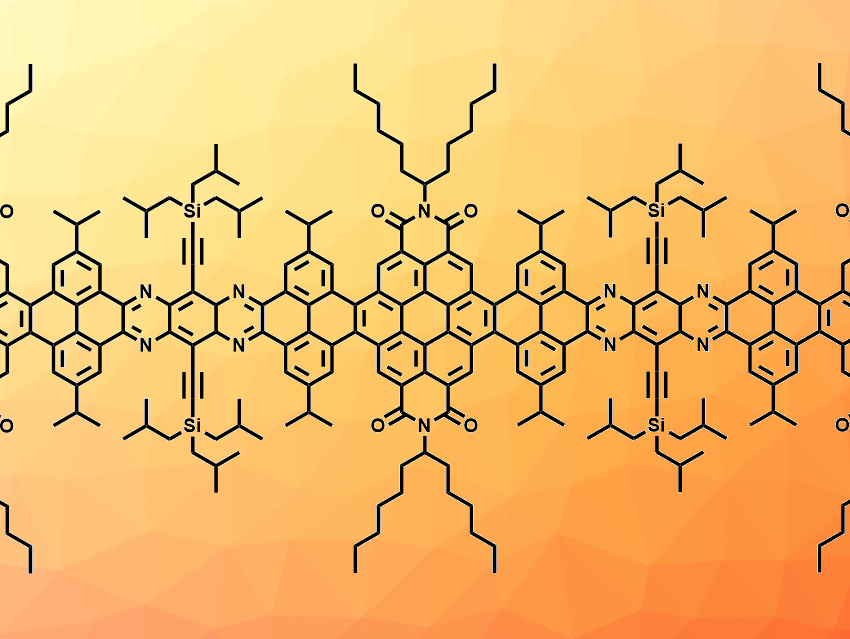
Aurelio Mateo-Alonso, University of the Basque Country, Donostia-San Sebastian, Spain, and Ikerbasque, Basque Foundation for Science, Bilbao, Spain, and colleagues have synthesized three molecular nanoribbons with 13, 33, and 53 linearly fused rings in their backbone, respectively (called NR-13, NR-33, and NR-53, latter pictured below). The nanoribbons were prepared from amino-functionalized benzene or benzo[c][1,2,5]thiadiazole units, dibromo-perylene-tetracarboxy bisimides, and pyrene building blocks, which were combined to form larger units. These building blocks were then fused via imine-type cyclocondensation reactions that form the connecting pyrazine rings.
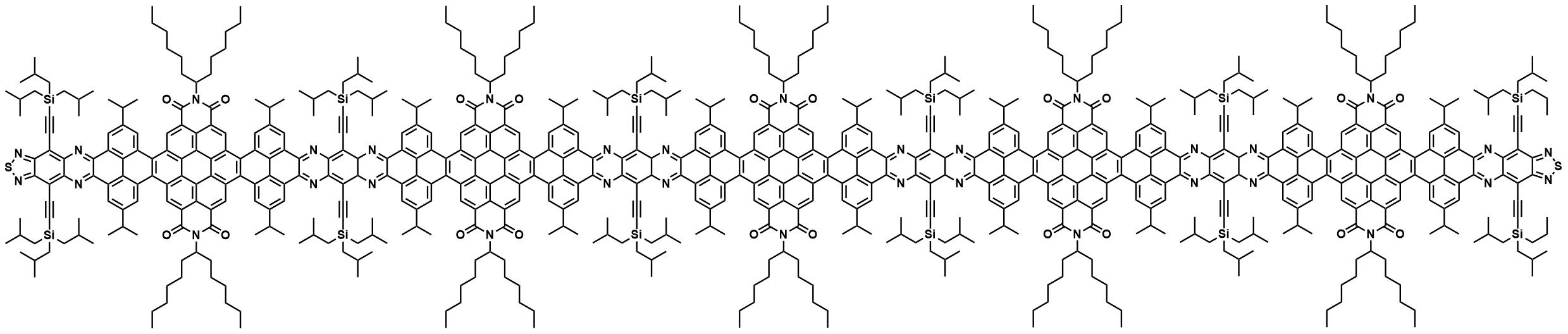
All three synthesized nanoribbons show red fluorescence, have twisted aromatic frameworks, and show high solubilities in common organic solvents, which allowed their separation via column chromatography and a thorough characterization. The lengths of NR-33 with 8.0 nm and NR-53 with 12.9 nm exceed the previous length record for molecular nanoribbons, which was 7.7 nm for a system with 30 linearly fused rings. NR-53, with 322 atoms in its conjugated aromatic core (C296N24S2), also sets a new record as the largest synthetic nanographene.
- Twisted Molecular Nanoribbons with up to 53 Linearly-Fused Rings,
Rajeev K. Dubey, Manuel Melle-Franco, Aurelio Mateo-Alonso,
J. Am. Chem. Soc. 2021.
https://doi.org/10.1021/jacs.1c01849