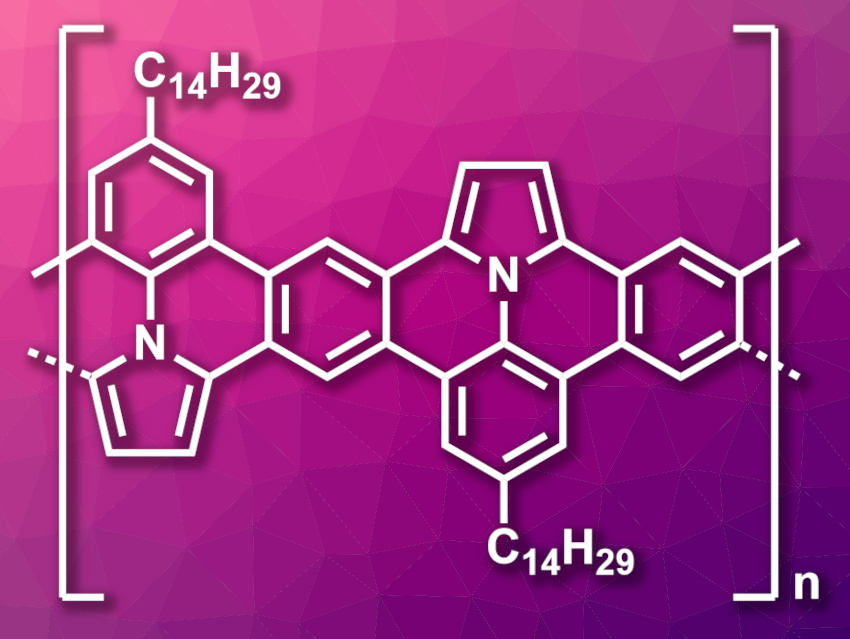
Polycyclic aromatic hydrocarbons and graphene nanoribbons (GNRs) can be modified using heteroatoms to tailor their optical and electronic properties for certain applications. Nitrogen atoms, for example, can be introduced in the form of pyrrole units. There are several methods for the introduction of pyrrole units in PAHs, and there have been examples of the on-surface synthesis of GNRs containing pyrrole units. However, the solution-phase preparation of pyrrole-embedded GNRs had not been reported so far.
Jean-François Morin, Université Laval, Québec, Canada, and colleagues have synthesized both linear and helical graphene nanoribbons containing pyrrole units (linear variant pictured) using a photochemical cyclodehydrochlorination reaction. The team started from 1-(4-tetradecyl-2,6-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)-1H-pyrrole, which was converted to polymeric precursors for the desired linear or helical GNRs using a Suzuki–Miyaura polymerization with 1,4-dichloro-2,5-diiodobenzene or 2,3-dichloro-1,4-diiodobenzene, respectively. The precursors were dissolved in decalin and then heated to 100 °C and irradiated with UV light to induce the photochemical cyclodehydrochlorination reaction and give the desired GNRs.
The team obtained the linear and helical pyrrole-embedded GNRs as dark brown and dark orange solids, respectively. According to the researchers, the linear variant is the first example of a fully planar N-substituted polypyrrole. Its structure provides an increased conductivity. Both GNRs could have applications in fluorescence sensing.
- Pyrrole-Embedded Linear and Helical Graphene Nanoribbons,
Dandan Miao, Vanessa Di Michele, Félix Gagnon, Cyril Aumaître, Andrea Lucotti, Mirella Del Zoppo, Frédéric Lirette, Matteo Tommasini, Jean-François Morin,
J. Am. Chem. Soc. 2021.
https://doi.org/10.1021/jacs.1c05616