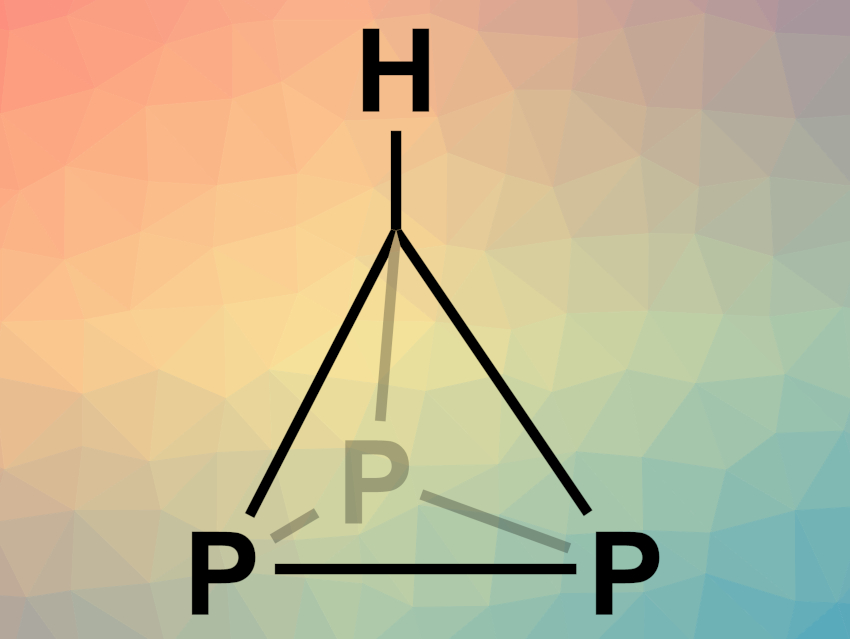
Tetrahedrane, a hypothetical tetrahedral hydrocarbon with the formula C4H4, has not been synthesized so far due to its instability and high strain. However, derivatives of this compound can be accessed. Exchanging atoms in the carbon core or using bulky substituents are two possible approaches for this. In phosphatetrahedranes, for example, carbon vertices are replaced with phosphorus atoms.
Christopher C. Cummins, Massachusetts Institute of Technology (MIT), Cambridge, MA, USA, and colleagues have synthesized the parent triphosphatetrahedrane HCP3 (pictured). The team reacted [Na(THF)3][P3Nb(ODipp)3] (Dipp = 2,6-diisopropylphenyl), INb(ODipp)3(THF), and bromodichloromethane in thawing tetrahydrofuran (THF). The Nb complex INb(ODipp)3(THF) abstracts a halogen atom from the bromodichloromethane, forming a radical. This carbon-centered radical can react with the niobatriphosphatetrahedrane [P3Nb(ODipp)3]– via the transfer of a P3 unit and form the desired triphosphatetrahedrane.
The team obtained HCP3 in a yield of 31 % (NMR internal standard yield). The compound is volatile. It was found not to be stable as a pure substance, but is stable in dilute THF solutions for some time. According to the researchers, the product might be useful for the synthesis of new organophosphorus compounds.
- Alleviating Strain in Organic Molecules by Incorporation of Phosphorus: Synthesis of Triphosphatetrahedrane,
Martin-Louis Y. Riu, Mengshan Ye, Christopher C. Cummins,
J. Am. Chem. Soc. 2021.
https://doi.org/10.1021/jacs.1c07959