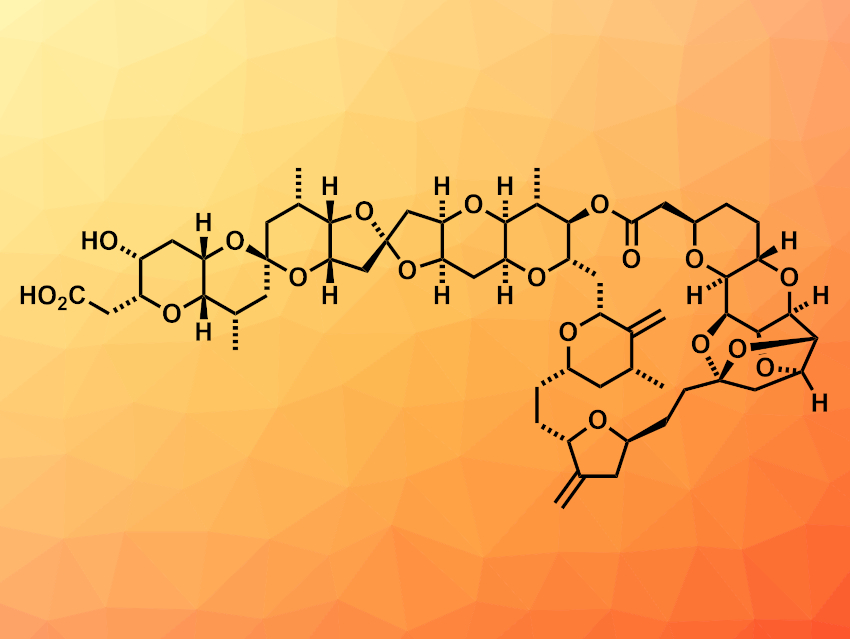
Norhalichondrin B (pictured) is a member of the halichondrin family of natural products. These compounds have complex structures and show biological activity, which makes them challenging and interesting targets for total synthesis. Existing syntheses of norhalichondrin B require a very large number of steps (at least 37 steps; longest linear sequence).
K. C. Nicolaou, Rice University, Houston, TX, USA, and colleagues have developed a highly convergent total synthesis of norhalichondrin B that requires only 25 steps. The team started from readily available tri-O-acetyl-D-glucal. They used a “reverse” strategy to construct cyclic ethers, in which C–O bonds are formed first via a Nicholas etherification reaction, followed by C–C bond formations. The researchers first constructed two large fragments with similar complexity: The fragments have 31 carbon atoms each and contain eight rings and six rings, respectively.
The two large fragments were coupled and converted to the desired product in five steps. According to the team, the route might allow further improvement in yields and number of steps, which could be helpful for the synthesis of other members of the halichondrin family, as well as analogues.
- A Highly Convergent Total Synthesis of Norhalichondrin B,
K. C. Nicolaou, Saiyong Pan, Yogesh Shelke, Qiuji Ye, Dipendu Das, Stephan Rigol,
J. Am. Chem. Soc. 2021.
https://doi.org/10.1021/jacs.1c10539