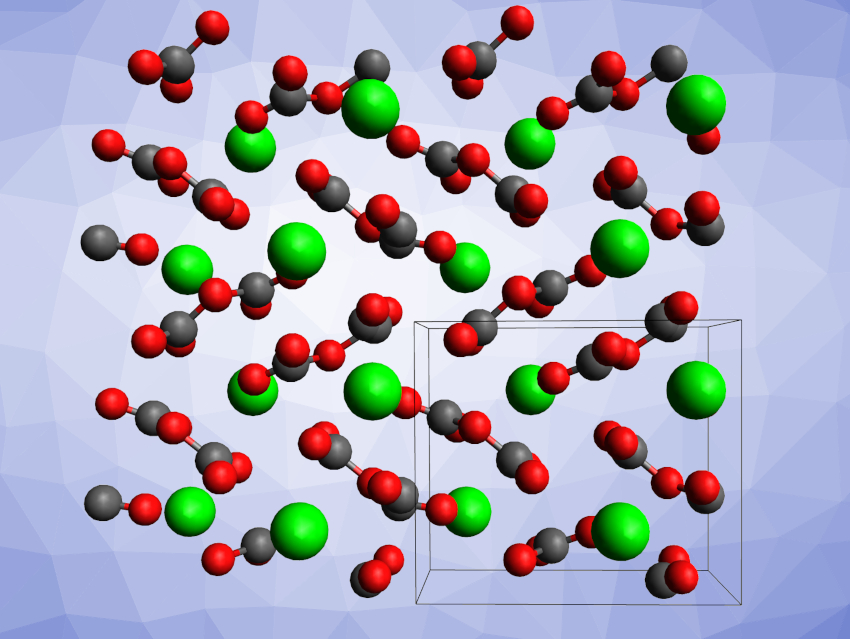
Inorganic carbonates usually contain a planar trigonal [CO3]2– unit. There are also carbonates with a tetrahedrally coordinated carbon atom with four surrounding oxygen atoms, i.e., carbonates with [CO4]4– units that may polymerize by corner-sharing. These can be obtained, for example, at very high pressures and temperatures. Pyrocarbonates, which contain [C2O5]2– units, can be found as pyrocarbonate esters in organic chemistry, but inorganic pyrocarbonates with isolated [C2O5]2– complex anions had not been reported so far.
Dominik Spahr, Goethe University Frankfurt, Frankfurt am Main, Germany, and colleagues have synthesized the inorganic pyrocarbonate salt Sr[C2O5], which contains isolated [C2O5]2– groups (structure pictured). The team reacted SrCO3 and CO2 at pressures of ca. 30 GPa and temperatures of ca. 2000 K in a laser-heated diamond anvil cell. The product was characterized using synchrotron-based single-crystal X-ray diffraction, which confirmed the presence of a novel pyrocarbonate with the composition Sr[C2O5].
The product crystallizes in the monoclinic space group P21/c. The [C2O5]2– groups can be considered to consist of two [CO3]2– units that share one oxygen atom. The team proposes that other inorganic pyrocarbonates, such as Mg[C2O5], Ca[C2O5], Ba[C2O5], and Pb[C2O5], might also be experimentally accessible.
- Sr[C2O5] is an Inorganic Pyrocarbonate Salt with [C2O5]2– Complex Anions,
Dominik Spahr, Jannes König, Lkhamsuren Bayarjargal, Victor Milman, Alexander Perlov, Hanns-Peter Liermann, Björn Winkler,
J. Am. Chem. Soc. 2022.
https://doi.org/10.1021/jacs.2c00351
Correction (February 10, 2022)
The article originally misidentified pyrocarbonates as pyroborates in the last sentence.