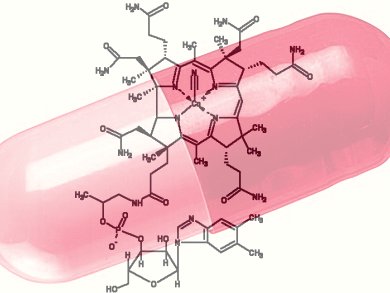
Derivatives of vitamin B12 are used in methyl group transfer in biological processes. These are as diverse as methionine synthesis in humans and CO2 fixation in acetogenic bacteria like Moorella thermoacetica. The seemingly straightforward reaction requires large, multimodular enzyme complexes that adopt multiple conformations to alternately activate, protect and perform catalysis on the reactive B12 cofactor. Crystal structures determined thus far have provided structural information for only fragments of these complexes.
Catherine L. Drennan, Massachusetts Institute of Technology (MIT), Cambridge, USA, and colleagues present X-ray crystal structures of a complete 220 kDa complex that contains all enzymes responsible for B12-dependent methyl transfer, namely the corrinoid iron–sulphur protein and its methyltransferase from the model acetogen M. thermoacetica. These structures provide the first 3-D depiction of all protein modules required for the activation, protection and catalytic steps of B12-dependent methyl transfer. They also capture B12 at multiple locations between its ‘resting’ and catalytic positions, allowing visualization of the dramatic protein rearrangements that enable methyl transfer and identification of the trajectory for B12 movement within the large enzyme scaffold.
The 3-D images reveal a multi-stage process involving what the researchers call an elaborate protein framework – a surprisingly complicated mechanism for such a simple, yet biologically essential reaction.
These findings are important to human health research and have potential for the development of new fuels that might become alternative renewable energy sources.
- Visualizing molecular juggling within a B12-dependent methyltransferase complex,
Yan Kung, Nozomi Ando, Tzanko I. Doukov, Leah C. Blasiak, Güneş Bender, Javier Seravalli, Stephen W. Ragsdale, Catherine L. Drennan,
Nature 2012.
DOI:10.1038/nature10916