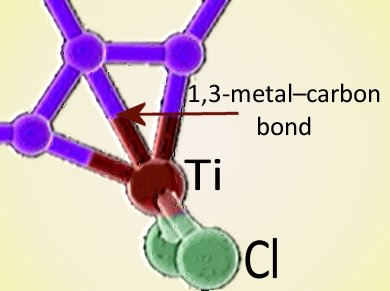
Cherumuttathu Suresh and Gernot Frenking, Philipps-Universität Marburg, Germany, have demonstrated the existence of 1,3-metal-carbon bonds based on CCD (charge-coupled device) X-ray structure analysis and density functional theory (DFT) calculations.
The new bonds form “edge complexes” in which the metal ion is coordinated at the V-shaped edges of olefinic and aromatic hydrocarbon moieties. The researchers point out that 1,2-metal-carbon bonds are well known, but their 1,3 counterparts were unprecedented. The molecules demonstrated by the team are planar, contain extended aromatic conjugation, and have small HOMO–LUMO gaps, which means they could have intriguing and potentially useful optoelectronic properties.
- 1,3-Metal–Carbon Bonding Predicts Rich Chemistry at the Edges of Aromatic Hydrocarbons,
Cherumuttathu H. Suresh, Gernot Frenking,
Organometallics 2013.
DOI: 10.1021/om301037a