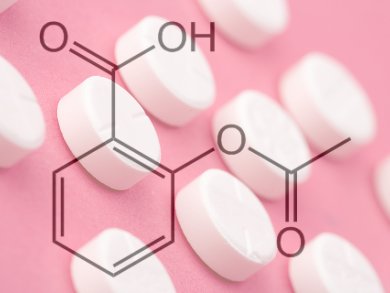
Aspirin is one of the most familiar and recognizable pharmaceuticals with 45,000 tonnes produced annually. While many organic chemists will have synthesized acetylsalicylic acid, fewer will be aware of the complexities of its crystalline and amorphous forms.
Researchers at New York University, USA, and colleagues at Curtin University, Perth, Australia, have shown that adding salicylic acid, the precursor of aspirin, to a melt can generate non-classical, helical crystals of aspirin. The discovery hints at the latent complications of trying to understand crystal forms when non-polyhedral structures of this kind are possible. The team used scanning electron microscopy, X-ray diffraction analysis, and optical polarimetry to obtain detailed images of the forms. Their next step will be to reveal which direction and how the growing crystallites choose a particular direction to twist and what effect salicylic acid concentration has on the mixed crystallization process.
- Twisted Aspirin Crystals,
Xiaoyan Cui, Andrew L. Rohl, Alexander Shtukenberg, Bart Kahr,
J. Am. Chem. Soc. 2013.
DOI: 10.1021/ja400833r