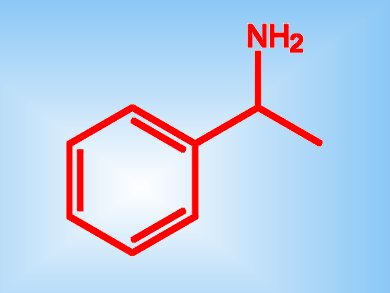
Optically pure amines are key chiral auxiliaries and intermediates in the pharmaceutical and agrochemical industries. Many synthetic procedures for chiral amines are scope limited and use environmentally harmful reagents; however, chemoenzymatic dynamic resolution seems the most versatile.
Rodrigo O. M. A. de Souza and colleagues, Federal University of Rio de Janeiro, Brazil, have developed a method for the kinetic resolution of (+/-)-1-phenylethylamine (pictured) that uses continuous-flow conditions. Starting from the racemic amine, reaction with a supported enzyme – Candida Antarctica lipase B immobilized on Novozyme 435 – and ethyl acetate gives a pure chiral amide product with an enantiomeric ratio >200.
This low-cost procedure performs well with short reaction times (40 m), high conversion rates, mild temperatures (50 °C), and reusable catalyst (at least 9 times without loss of activity) that make it suitable for automation and scale-up.
- Ethyl Acetate as Acyl Donor on the Continuous Flow Kinetic Resolution of (+/-)-1-phenylethylamine Catalyzed by Lipases,
Rodrigo O. M. A de Souza, Leandro S. M. Miranda, Amanda S. de Miranda,
Org. Biomol. Chem. 2013.
DOI: 10.1039/C3OB40437D