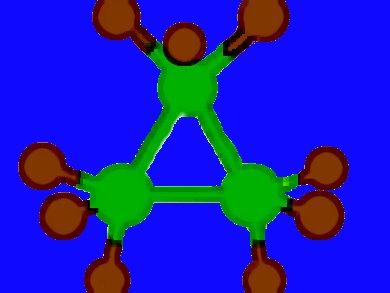
Eluvathingal D. Jemmis, Indian Institute of Science, Bangalore, India, and a colleague from India found that P3F92− or (PF3)32− is an all-pseudo π* 2π-aromatic system. Its aromaticity comes solely from the interaction of pseudo-π* MOs. The dianion forms the first example of a three membered ring with all the vertices constituted by pentacoordinate phosphorus.
Since 1950 the analogy between CO and PF3 has been known. The similarity is due to the fact that CO has a low-lying π* MO that is localized more on C, and PF3 has a low-lying pseudo-π* MO that is localized more on P. In 1997 the deltate anion, (C3O3)2−, was discovered, however, its PF3 analogue remained unnoticed until now.
The researchers assume that (PF3)32− can be synthesized via a procedure similar to the synthesis of C3O32− from CO via dissolving metal reduction. The heavier analogues, (AsF3)32− and (SbF3)32−, are more weakly bound and may be harder to realize, they say.
- P3F92−: An All-Pseudo-π* 2π-Aromatic,
Chakkingal P. Priyakumari, Eluvathingal D. Jemmis,
J. Am. Chem. Soc. 2013.
DOI: 10.1021/ja408308g