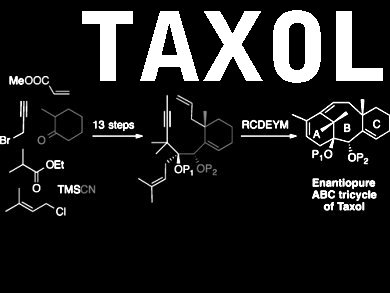
Taxol and its derivatives are very potent anticancer treatments, which are widely used to treat a variety of cancers including ovarian, bladder and lung cancer. Taxol has a complex, tricyclic core, and although a number of total syntheses have been developed, its significance as a drug warrants additional studies.
Joëlle Prunet and colleagues at the University of Glasgow, UK, and the Ecole Polytechnique, France, have developed an efficient and selective synthesis of the tricyclic core. The key step of the synthesis is one in which all three rings are formed one after another in a single operation to form the core, a cascade metathesis reaction. By fine-tuning the protecting groups and the stereochemistry of the starting materials, a three-ringed product with the required stereochemistry is produced.
Overall, the core was synthesized in 14 steps in 11 % yield from the readily available starting material ethyl isobutyrate. Efforts to complete the synthesis of Taxol from the synthesized core are now underway.
- Highly Efficient Synthesis of the Tricyclic Core of Taxol by Cascade Metathesis,
Aurélien Letort, Rémi Aouzal, Cong Ma, De-Liang Long, Joëlle Prunet,
Org. Lett. 2014.
DOI: 10.1021/ol501304j