“Ultra-thin and flexible” are desirable features for many electronic systems such as screens, smart phones, notebooks, and televisions. Energy storage devices that support these features are thus needed. Conventional batteries use liquid electrolytes, requiring a tricky sealing process to avoid any leakage as the electrolyte solutions can be toxic and corrosive.
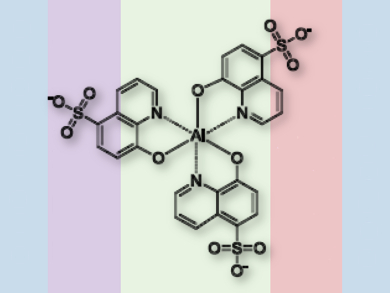
Youngkyoo Kim and colleagues, Kyungpook National University, Daegu, South Korea, designed a small molecular metal-chelate complex, tris(8-hydroxyquiinoline-5-sulfonic acid) aluminum (AlQSA3, pictured). It is easily produced in a simple one-step-synthesis from aluminum triisopropoxide and 8-hydroxyquinoline-5-sulfonic acid. The molecule’s three sulfonic acid groups secure protons for energy storage.
The complex is water-soluble, which comes in handy since aqueous solutions of the complex form thin films during drying. AlQSA3 films were wet-coated on an indium-tin oxide (ITO) electrode and dried, then another ITO substrate was stacked on top, forming all-solid state electric double layer capacitors. In a dried state, AlQSA3 exhibits high ion conductivity and the capacitors show stable performance even after repeated bending.
- Aqueous Solution-Processable Small Molecular Metal-Chelate Complex Electrolyte for Flexible All-Solid State Energy Storage Devices,
Hyena Lee, Jaehoon Jeong, Hwajeong Kim, Youngkyoo Kim,
Adv. Energy Mat. 2015.
DOI: 10.1002/aenm.201500402