Delivering chemotherapy agents directly and selectively to tumor cells allows the use of higher doses with less side effects and better overall results. Nanoparticles can be used to achieve this, but have so far been limited to passive mechanisms that predominantly target tumors which undergo rapid angiogenesis (the formation of new blood vessels).
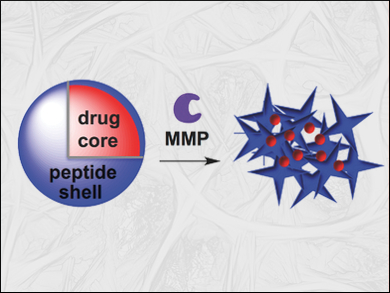
Nathan C. Gianneschi, University of California, San Diego, La Jolla, CA, USA, and colleagues have developed nanoparticles loaded with a chemotherapeutic compound which actively target tumors. The compound, paclitaxel, is a commonly used chemotherapy drug. The team modified it with a norbornene substituent, which both inactivated the drug and allowed it to be co-polymerized with a similarly modified peptide using ring-opening metathesis polymerization (ROMP).
The co-polymer assembled into nanoparticles with a paclitaxel core and a peptide shell. The peptide shell served as a substrate for enzymes called matrix metalloproteinases (MMPs). These enzymes are expressed in a range of cancer types, usually destroy membranes and allow cancer cells to escape and metastasize. Upon contact with the enzymes, the nanoparticles’ shell is disintegrated and their drug cargo released and activated.
In a study on mice, the team was able to use a paclitaxel dose 16 times higher than the usual clinical formulation without noticably higher toxicity. The researchers hope that this approach can be generalized for other therapeutic agents which can be modified with a norbornene unit.
- Therapeutic Enzyme-Responsive Nanoparticles for Targeted Delivery and Accumulation in Tumors,
Cassandra E. Callmann, Christopher V. Barback, Matthew P. Thompson, David J. Hall, Robert F. Mattrey, Nathan C. Gianneschi,
Adv. Mater. 2015.
DOI: 10.1002/adma.201501803