N-heterocyclic carbenes (NHCs) with bulky substituents have applications in stabilizing main-group compounds with low oxidation states and in transition-metal catalysis. An alternative to the common bulky, yet flexible, alkyl and aryl substituents are icosahedral carboranes.
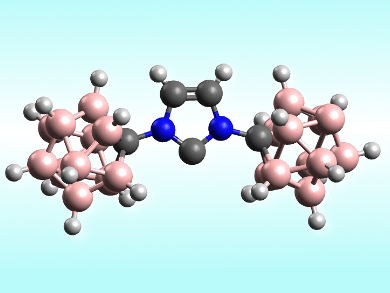
Vincent Lavallo, University of California, Riverside, USA, and colleagues have synthesized the first examples of carboranyl-NHC complexes with transition metals. They used both a monoanionic NHC with one carborane substituent and a dianionic symmetrical NHC with two carborane groups (pictured) and synthesized the corresponding Au(I) carbene complexes.The team reacted ClAuSMe2 and the lithium salts of the carboranyl-NHCs in C6H5F as a solvent and obtained the corresponding zwitterionic and monoanionic gold complexes [Au(NHC)nSMe2]m.
The products were characterized by 1H, 13C, and 11B NMR spectroscopy, as well as X-ray crystallography. The researchers also calculated their percent buried volume (%Vbur) as a measure for the ligands’ steric bulk. The mono- and dicarboranyl-NHCs had %Vbur-volumes of 44.6 and 47.2, respectively, close the the bulkiest previously known NHC ligands.
The team is currently investigating the complexes’ catalytic activity and exploring the synthesis of even more sterically demanding carboranyl-NHCs.
- Anionic and Zwitterionic Carboranyl N-Heterocyclic Carbene Au(I) Complexes,
Steven P. Fisher, Ahmad El-Hellani, Fook S. Tham, Vince Lavallo,
Dalton Trans. 2016.
DOI: 10.1039/C6DT00551A