The heavier analogues of alkynes, such as (RGe)2, are usually stabilized by sterically demanding ligands, and have bent geometries, in contrast to the linear alkynes. They are often highly reactive towards small molecules such as H2, as well as amines, boranes, alkenes, and alkynes. However, there have been few reports of such compounds as ligands in transition metal complexes.
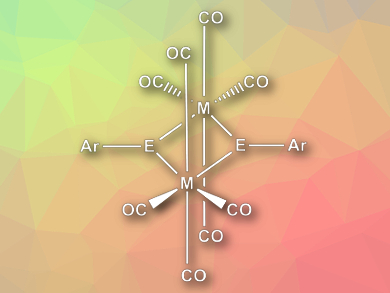
Heikki M. Tuononen, University of Jyväskylä, Finland, Philip P. Power, University of California, Davis, USA, and colleagues have investigated the reactivity of group 14 dimetallynes of the type (ER)2 (E = Ge, Sn) towards transition-metal carbonyl complexes M(CO)6 (M = Cr, Mo, W). Instead of simple adducts, the team found dinuclear metal complexes (pictured) when the reactants were irradiated with ultraviolet light. These complexes result from the cleavage of the formal triple bond between the group 14 elements and the successive complexation of two M(CO)4 fragments.
The researchers characterized the compounds using NMR spectroscopy and X-ray crystallography. The complexes have a planar rhomboid M2E2 core, with octahedral coordination of the transition metals and trigonal planar coordination of the group 14 elements. The team performed quantum chemical calculations to investigate the bonding in the complex and found evidence for four single bonds in the M2E2 core. According to the researchers, computational efforts to characterize the mechanism of the complexes’ formation are ongoing.
- Cleavage of Ge–Ge and Sn–Sn Triple Bonds in Heavy Group 14 Element Alkyne Analogues (EAriPr4)2 (E = Ge, Sn; AriPr4= C6H3-2,6(C6H3-2,6-iPr2)2) by Reaction with Group 6 Carbonyls,
Madison L. McCrea-Hendrick, Christine A. Caputo, Jarno Linnera, Petra Vasko, Cory M. Weinstein, James C. Fettinger, Heikki M. Tuononen, Philip P. Power,
Organometallics 2016.
DOI: 10.1021/acs.organomet.6b00519