Removing CO2 from the atmosphere and transforming it into species that can be used as a chemical feedstock is an important challenge for chemistry. One approach is the reduction of CO2 to CO or hydrocarbons. A number of electrocatalysts for this type of reaction have been developed. Homogeneous nickel catalysts are particularly efficient and selective, however, they can be difficult to recover and reuse.
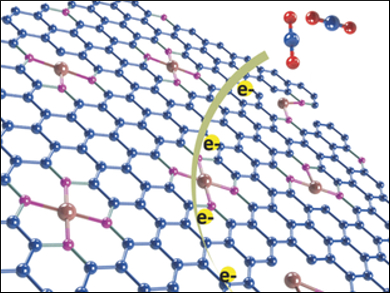
Kazuhito Hashimoto, Kazuhide Kamiya, University of Tokyo, Japan, and colleagues have developed a heterogeneous nickel-nitrogen-doped graphene catalyst which effectively reduces CO2 to CO. The team heat-treated nickel-pentaethylenehexamine and graphene oxide (GO) under an inert atmosphere at 900 °C to produce Ni-N-modified graphene (Ni-N-Gr).
The resulting material was characterized using transmission electron microscopy (TEM), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and extended X-ray absorption fine structure (EXAFS) measurements. The nickel atoms were found to be coordinated by nitrogen atoms and uniformly dispersed in the graphene sheets.
The modified graphene has a high electrocatalytic activity for CO2 reduction and good selectivity for the formation of CO. The researchers found that both nickel and nitrogen are necessary for the high catalytic activity, and they conclude that the Ni-N sites are the catalytically active center.
- Nickel-Nitrogen-Modified Graphene: An Efficient Electrocatalyst for the Reduction of Carbon Dioxide to Carbon Monoxide,
Panpan Su, Kazuyuki Iwase, Shuji Nakanishi, Kazuhito Hashimoto, Kazuhide Kamiya,
Small 2016.
DOI: 10.1002/smll.201602158