9,10-Diphenylanthracene fluoresces with a deep blue color and a very high quantum yield. This makes the compound a good candidate for organic light-emitting diodes (OLEDs). However, its tendency to decompose is a major drawback.
Emil J. W. List-Kratochvil, Humboldt-Universität zu Berlin and Helmholtz Zentrum Berlin für Materialien und Energie, Germany, Jan Freudenberg, Uwe H. F. Bunz, University of Heidelberg, Germany, and colleagues have stabilized anthracene by an encapsulation of its reactive core using different bridges. The team prepared a series of doubly bridged anthracenes (pictured below) and investigated their photostability. They found that attachment of the bridges via electron-withdrawing ester groups (example pictured above) improved the stability the most, while the bridge length and the nature of the bridge had a smaller impact. The ester bridges were installed via the fourfold esterification of a known diresorcinylanthracene with diacyl chlorides.
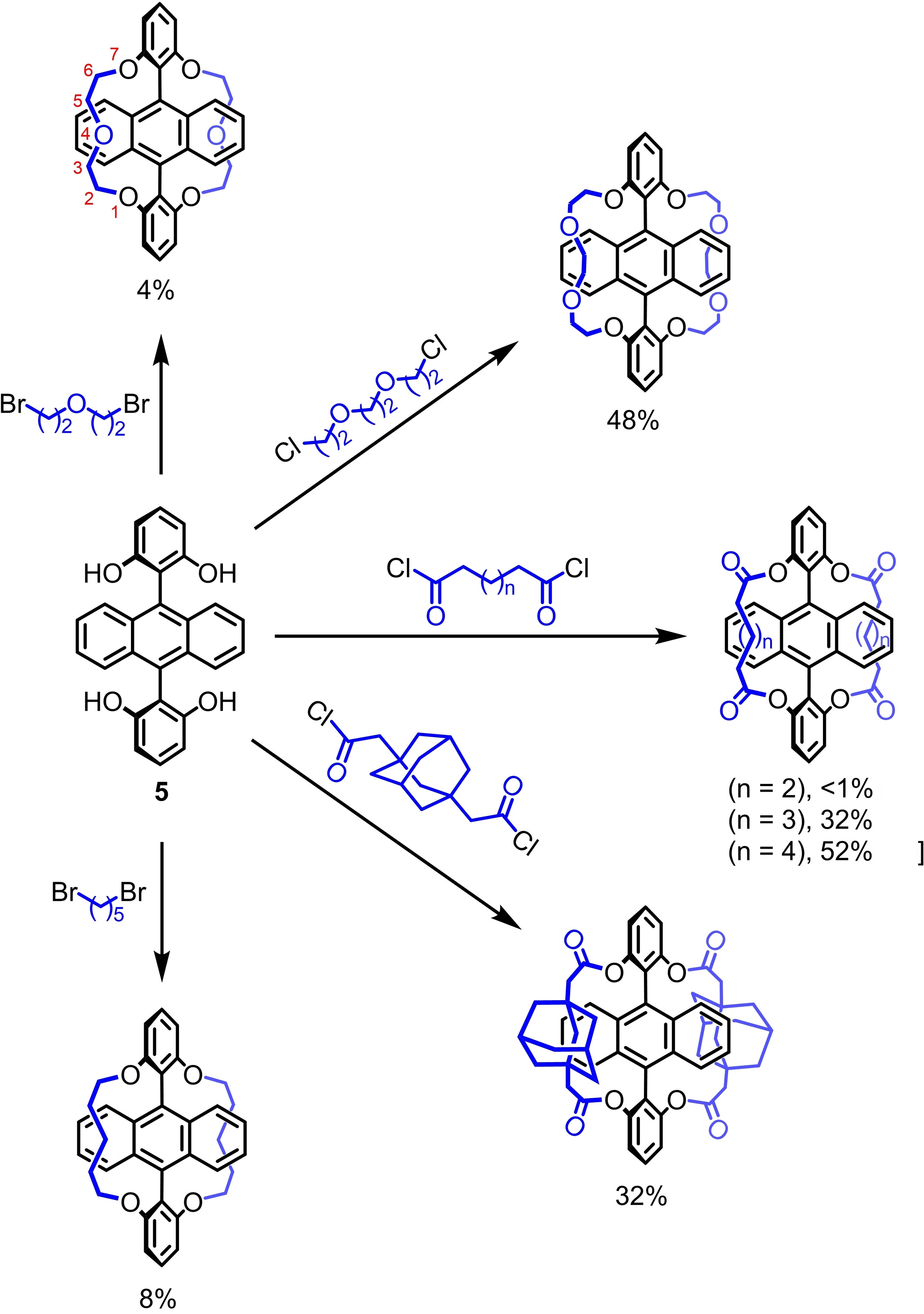
Upscaling of the synthesis of the most promising compounds enabled the fabrication of proof-of-principle OLEDs. Thus, anthracene can be stabilized at the molecular level without sacrificing its useful emitting properties, using a simple synthesis. Overall, “intramolecular encapsulation” might be a valuable stabilization technique both in fundamental research and in real-world applications.
- Doubly Bridged Anthracenes: Blue Emitters for OLEDs,
Philipp Ludwig, Jacob Mayer, Lukas Ahrens, Frank Rominger, Giovanni Ligorio, Felix Hermerschmidt, Emil J. W. List‐Kratochvil, Jan Freudenberg, Uwe H. F. Bunz,
Chem. Eur. J. 2023.
https://doi.org/10.1002/chem.202303037




