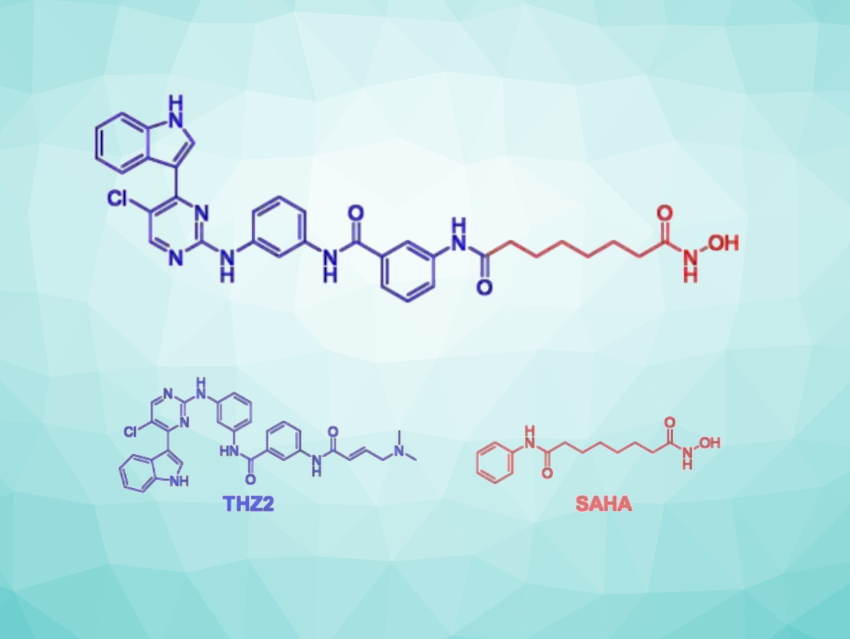
In cancer cells, the enzymes cyclin-dependent kinase 7 (CDK7) and histone deacetylase 1 (HDAC1) can be highly expressed and serve as useful drug targets. The over-activation of CDK7 leads to cell-cycle abnormalities, and HDAC1 regulates the expression of many tumor-associated genes. Therefore, the development of dual CDK7/HDAC1 inhibitors may be a promising anticancer strategy.
Lan Zhang, Southwest Jiaotong University, Chengdu, China, and colleagues have combined the pharmaceutically active units of THZ2 (a CDK7 inhibitor) and SAHA (an HDAC inhibitor, both pictured above) to design novel dual-target CDK7/HDAC1 inhibitors. A suitable linker allows effective simultaneous inhibition of both targets.
The THZ2-type fragment was prepared starting from indole, which was reacted with 2,4,5-trichloropyrimidine, benzenesulfonyl chloride, m-phenylenediamine, and m-nitrobenzoyl chloride, followed by a reduction. The resulting intermediate was reacted with various carboxylic acid methyl esters—serving as precursors for the linker/SAHA-type unit—and an alkaline NH2OH methanol solution to obtain the target compounds. The team tried a variety of flexible and rigid linkers, including aliphatic chains of various lengths and aromatic heterocycles.
Ultimately, the researchers obtained a potent dual-target CDK7/HDAC1 inhibitor with broad-spectrum anticancer effects. Pharmacological studies verified that the best compound arrests the cell cycle, induces apoptosis, and inhibits cell migration in colorectal cancer cells. Overall, the work provides a new strategy for cancer therapy and facilitates further development of dual-target drug candidates.
- Discovery of a dual‐target inhibitor of CDK7 and HDAC1 that induces apoptosis and inhibits migration in colorectal cancer,
Yao Chen, Shuangqian Zhang, Zhijia Li, Bo Yin, Yi Liu, Lan Zhang,
ChemMedChem 2023.
https://doi.org/10.1002/cmdc.202300281