Highly air- and moisture-sensitive N-heterocyclic carbenes (NHCs) are often prepared in situ by treatment of imidazolium precursors with strong alkali reagents. This synthetic method can lead to formation of inactive NHC dimers and to the destruction of porous supports. Moreover, it is often difficult to recover and reuse NHCs or their precursors.
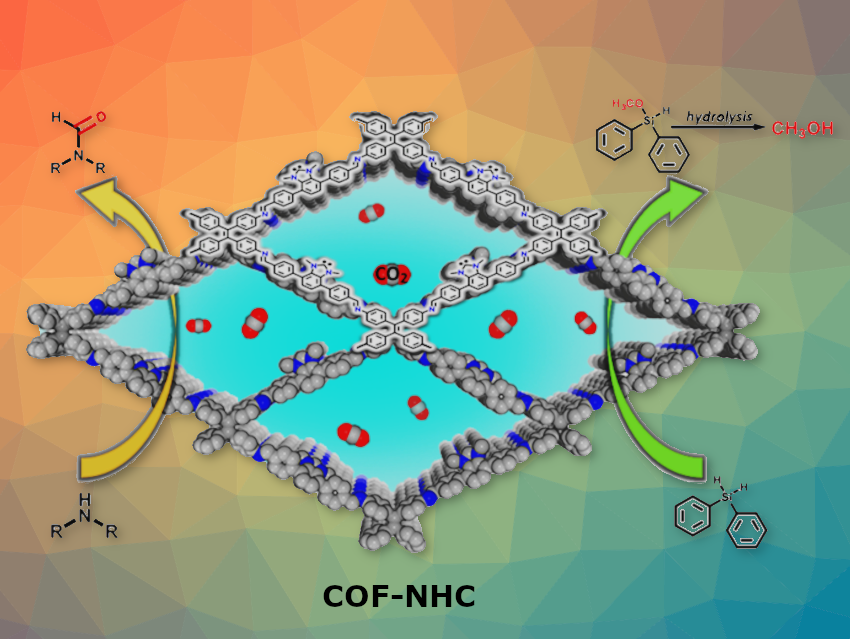
Rong Cao, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou and colleagues have synthesized a CO2-masked NHC-functionalized covalent organic framework (COF) for highly efficient catalytic CO2 conversion. These are the first stable CO2-masked N-heterocyclic carbenes (NHCs) grafted on a porous crystalline COF.
The imidazole-functionalized Im-COF-1 was synthesized by condensing 4,7-bis(4-formylbenzyl)-1-methyl-1H-benzimidazole and 4,4′,4”,4”’-(ethene-1,1,2,2-tetrayl)-tetraaniline. The researchers then reacted the imidazole-functionalized Im-COF-1 with dimethyl carbonate at 120 °C for 24 h, forming NHC-CO2-COF.
The well designed porous COF-NHC-CO2 shows superior activity and selectivity in the hydrosilylation of CO2 and N-formylation of amines with CO2 to produce methanol and formamides, respectively.
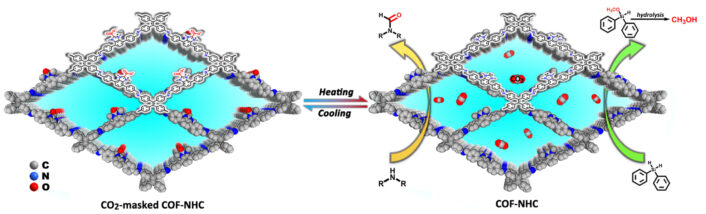
On application of heat, the CO2 moiety was released in situ to form free NHC single active sites. The NHC sites were highly active in catalytic hydrosilylation and N-formylation reactions with CO2. Moreover, exposure to a CO2 atmosphere regenerated the NHC-CO2 moieties.
- A CO2‐Masked Carbene Functionalized Covalent Organic Framework for Highly Efficient Carbon Dioxide Conversion,
Chang He, Duan-Hui Si, Yuan-Biao Huang, Rong Cao,
Angew. Chem. Int. Ed. 2022.
https://doi.org/10.1002/anie.202207478