Hydrocarbon “belts” have interesting structures and could be useful, e.g., as synthetic macrocyclic host molecules. Such belt-type molecules with non-hexagonal rings are rare. The synthesis of fully conjugated “zigzag”-type hydrocarbon belts can be challenging because of the molecular strain and the ground-state open-shell electronic configuration that is predicted by theoretical calculations.
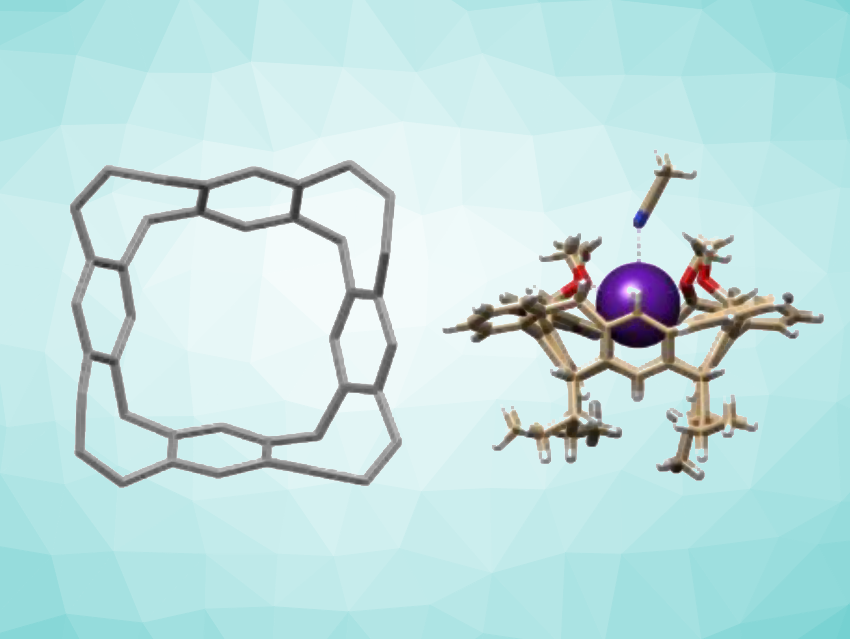
Shuo Tong, Mei-Xiang Wang, Tsinghua University, Beijing, China, and colleagues have synthesized unprecedented zigzag-type hydrocarbon belts which contain functionalized eight-membered rings (simplified structure pictured above on the left). The team started from calix[4]arene derivatives, which were iodinated. The resulting iodoarenes underwent fourfold Suzuki-Miyaura coupling reactions with different arylboronic acids to install additional aryl rings. A fourfold intramolecular Friedel-Crafts acylation reaction then gave ketone-containing, belt-shaped compounds, i.e., tetrabenzobelt[4]arene[4]cyclooctatrienones.
The ketone-containing intermediates were then further transformed. A ketone reduction with LiAlH4 or a nucleophilic addition using alkynyl lithium reagents provided the corresponding tetrahydroxylated belts. The researchers found that enantiopure molecular belts exhibit circularly polarized luminescence (CPL) activity. In addition, some of the functionalized belt compounds can serve as selective host molecules to bind cesium ions (example complex pictured above on the right). Overall, the work provides a useful method to construct hydrocarbon belt structures with non-hexagonal rings as well as new functional chiroptical materials and adsorbents for Cs+.
- Functionalized Hydrocarbon Belts: Synthesis, Structure and Properties,
Yi Peng, Shuo Tong, Yang Zhang, Mei-Xiang Wang,
Angew. Chem. Int. Ed. 2023.
https://doi.org/10.1002/anie.202302646