Developing theranostic probes for noninvasive bone imaging and the treatment of bone diseases is an interesting research target. Such probes could be used to monitor, e.g., bone metabolism, growth, metastases, and drug delivery, while also allowing for high-efficacy therapies.
Conventional bone imaging probes often have bisphosphonates as bone-targeting units and usually emit visible up to near-infrared (NIR) photoluminescence in the so-called first window (NIR-I). Such probes have drawbacks such as a long half-life, difficult clearance in vivo, poor spatial resolution, and weak tissue penetration. High-resolution noninvasive bone imaging and efficient therapies for some bone diseases such as rheumatoid arthritis remain challenging targets—especially with a theranostic probe that is suitable for both.
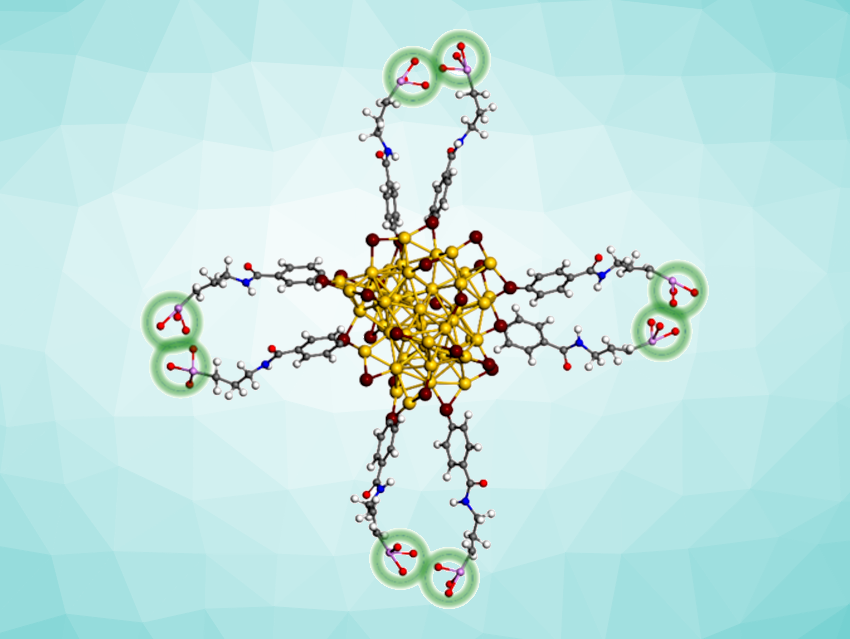
Xun Yuan, Qingdao University of Science and Technology, China, and colleagues have developed ultrasmall gold nanoclusters with a core size of less than 2 nm that can serve as highly precise and noninvasive theranostic probes. These probes enable high-resolution bone-targeted photoluminescence imaging in the second near-infrared window (NIR-II) and enhanced rheumatoid arthritis therapy. The team used NIR-II emitting Au44MBA26 nanoclusters (MBA = 4-mercaptobenzoic acid) and modified their surface with bone-targeting 3-amino-propane-1-phosphate molecules (APP).
The researchers confirmed that the phosphorylation improved the bone-targeting ability and allowed in vivo NIR-II photoluminescence bone imaging. Additionally, the team demonstrated that the nanocluster probe exhibited low cytotoxicity and high biocompatibility, and that it showed anti-inflammatory activity against rheumatoid arthritis in a rat model.
- Phosphorylation of NIR‐II emitting Au nanoclusters for targeted bone imaging and improved rheumatoid arthritis therapy,
Ge Yang, Kang Liu, Yaru Wang, Xinxin Pan, Jingrun Ye, Yue Li, Fanglin Du, Ting Feng, Xun Yuan,
Aggregate 2023.
https://doi.org/10.1002/agt2.435