The development of new fluorescent molecules is an active area of research due to their broad applicability and contributions to various scientific disciplines. Boron-dipyrromethene dyes (BODIPYs) stand out as a class of chromophores that offer highly tunable optoelectronic properties. However, much less is known about BODIPY analogues with heavier main-group elements as the central atom.
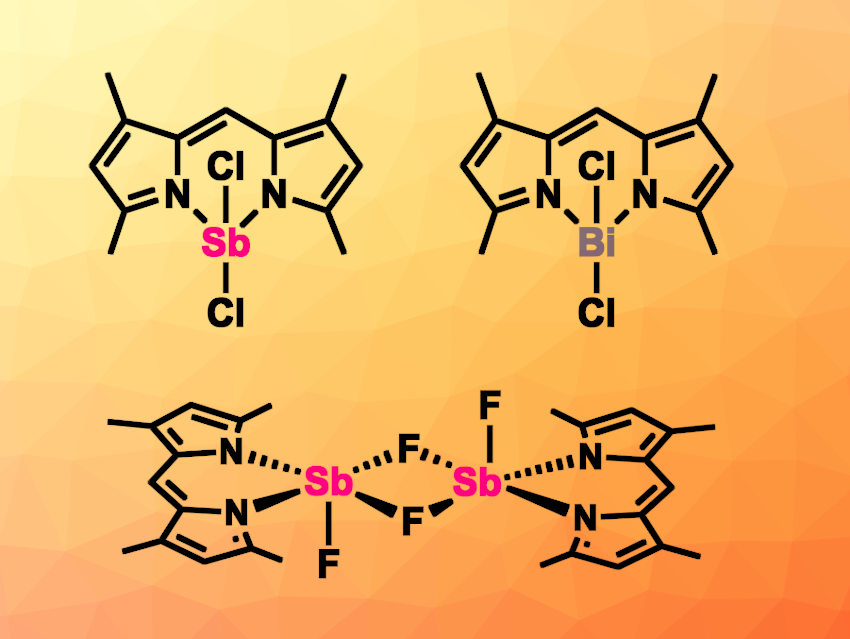
Alessandro Bismuto, University of Göttingen and University of Bonn, both Germany, and colleagues have reported the synthesis and characterization of antimony- and bismuth-based dipyrromethene complexes, celled SBDIPY and BIDIPY. The team added a tetrahydrofuran (THF) solution of different dipyrrin ligands and lithium bis(trimethylsilyl)amide (LiHMDS) to a THF solution of SbF3, SbCl3, or SbBr3, to obtain SBDIPY complexes (example pictured above on the top left). The product obtained from SbF3 has a dimeric structure (pictured above in the bottom row). According to the researchers, this is the first direct analogue of BODIPY containing fluoride handles. To obtain BIDIPYs, the team reacted BiCl3 and sodium dipyrrinato salts in THF.
The researchers investigated the luminescence features of the complexes and found that replacing the boron in BODIPY with a heavier central atom has only minimal influence on the main electronic transition. The compounds have similar luminescence properties, however with notably lower stability, especially for bismuth.
- Replacing the BO in BODIPY: unlocking the path to SBDIPY and BIDIPY chromophores,
André Korzun, Stefano Crespi, Christopher Golz, Alessandro Bismuto,
Chem. Sci. 2023.
https://doi.org/10.1039/d3sc01493b