Organic batteries could be useful for storing electricity in a more sustainable way compared with conventional metal-based battery electrodes. Redox-active organic molecules can offer high charge/discharge capacities, but their stability and the achievable voltages are limited so far.
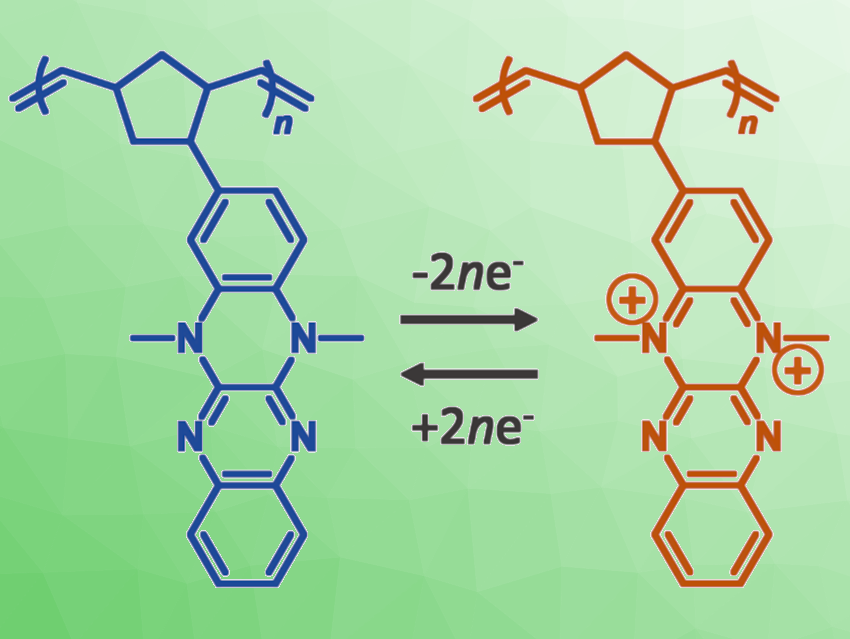
Kenichi Oyaizu, Waseda University, Tokyo, Japan, and colleagues have synthesized a new 4 V-class cathode-active polymer for lithium-ion batteries. The polymer (pictured) has a polynorbornene backbone, which was substituted with a quadruply fused aromatic heterocycle, i.e., dimethylfluoflavin. The polymer was synthesized via ring-opening metathesis polymerization.
The synthesized dimethylfluoflavin-functionalized polymer offered high redox potentials of 3.5 and 4.1 V vs. Li/Li+. It also showed excellent cycle performance, with over 90 % capacity retention after 1,000 charge/discharge cycles. Other quadruply fused aromatic heterocycles also showed high potentials. The results could be helpful for the development of organic batteries with higher energy densities and improved stability, possibly leading to more sustainable energy storage.