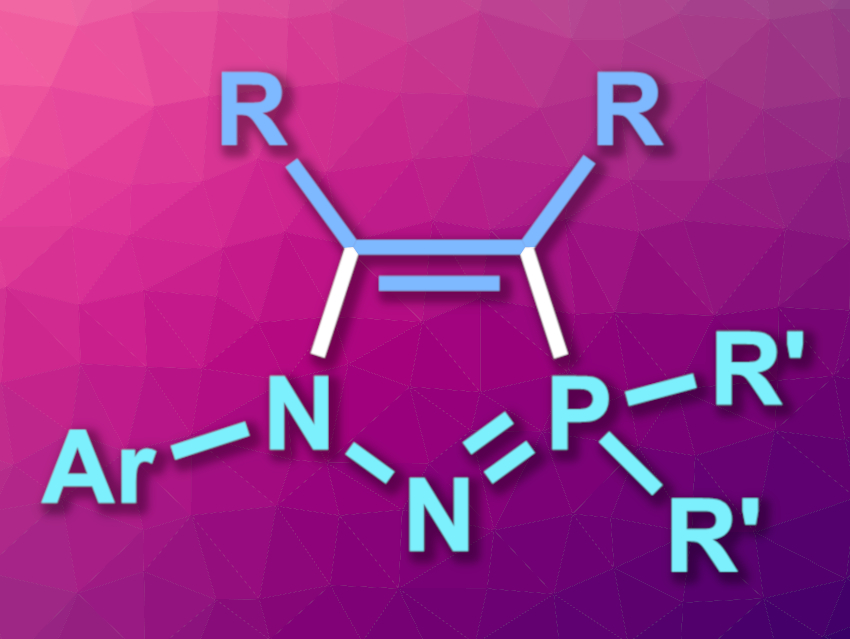
Iminophosphoranes (R3P=NR) have a basic nitrogen atom and can be useful, e.g., as ligands or organocatalysts. Cyclic iminophosphoranes with a more rigid structure can have applications, for example, in the selective recognition of anions or the stabilization of reactive species.
Christopher C. Cummins, Massachusetts Institute of Technology (MIT), Cambridge, USA, and colleagues have prepared a new class of heterocycles based on iminophosphoranes, N-heterocyclic iminophosphoranes (NHIPs, general structure pictured). These compounds have a similar structure to cyclic (alkyl)(amino)carbenes (CAACs), which are widely used as ligands.
The team first synthesized azophosphines (Ar–N=N–PR2). They were prepared either from N-aryl-N′-(trimethylsilyl)diazenes (Ar–N=N–SiMe3) via a reaction with R2PCl or from phosphinohydrazines (Ar–NH–NH–PR2) via an oxidation using benzoquinone derivatives. The azophosphines were then reacted with alkynes, i.e., either cyclooctyne or dimethylacetylene dicarboxylate, in a 1,3-dipolar cycloaddition in thawing benzene.
The corresponding N-heterocyclic iminophosphoranes were obtained in generally moderate to good yields. They are strongly basic and could have applications as ligands in transition-metal and main-group chemistry, as well as in catalysis.
- Introducing N-Heterocyclic Iminophosphoranes (NHIPs): Synthesis by [3 + 2] Cycloaddition of Azophosphines with Alkynes and Reactivity Studies,
Keita Tanaka, Martin-Louis Y. Riu, Brian Valladares, Christopher C. Cummins,
Inorg. Chem. 2022.
https://doi.org/10.1021/acs.inorgchem.2c01844