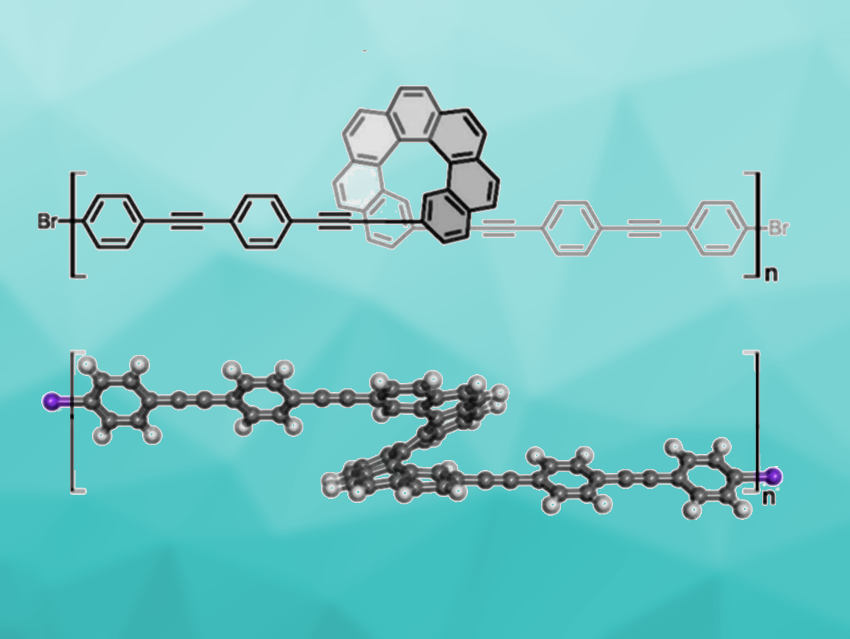
The bottom-up synthesis of nanomaterials can be useful for their controlled preparation—in particular, on-surface syntheses can allow the creation of covalently linked nanostructures that are inaccessible in solution. Helicenes are chiral polycyclic aromatic hydrocarbons (PAHs) with a screw-shaped structure. Oligomeric/polymeric helicene chains are interesting one-dimensional materials that combine chirality with an extended π-conjugated system. However, the maximum length of single helicenes and their analogues that can be synthesized is still limited. There are examples of oligomers/polymers with helicenes in their main chain, but so far, no main-chain helicene oligomers/polymers had been prepared by on-surface synthesis.
Oleksandr Stetsovych, Institute of Physics, Czech Academy of Sciences, Prague, Czech Republic, and colleagues have achieved an oligomerization of a racemic helicene derivative (2,17-bis(4-[(4-bromophenyl)ethynyl]phenylethynyl) heptahelicene), using an Ullmann coupling on an Au(111) surface (product pictured). The 1-ethynyl-4-(phenylethynyl)benzene units serve as spacers to prevent steric hindrance between the helicene monomers during oligomerization while maintaining π-conjugation.
The team started from a racemic 2,17-bis((triisopropylsilyl)ethynyl)heptahelicene, which was desilylated and reacted with 1-bromo-4-((4-iodophenyl)-ethynyl)benzene in a Sonogashira coupling. The resulting brominated monomers were deposited on an Au(111) surface and oligomerized by heating under ultrahigh vacuum conditions. The researchers characterized the products using scanning tunneling microscopy (STM), non-contact atomic force microscopy (nc-AFM), and theoretical calculations. Various heterochiral oligomers, from dimers up to octamers, were identified on the Au(111) surface.
- On‐Surface Synthesis of Helicene Oligomers,
Andrés Pinar Solé, Jiří Klívar, Michal Šámal, Irena G Stará, Ivo Starý, Jesús Mendieta-Moreno, Karl-Heinz Ernst, Pavel Jelínek, Oleksandr Stetsovych,
Chem. Eur. J. 2024.
https://doi.org/10.1002/chem.202304127