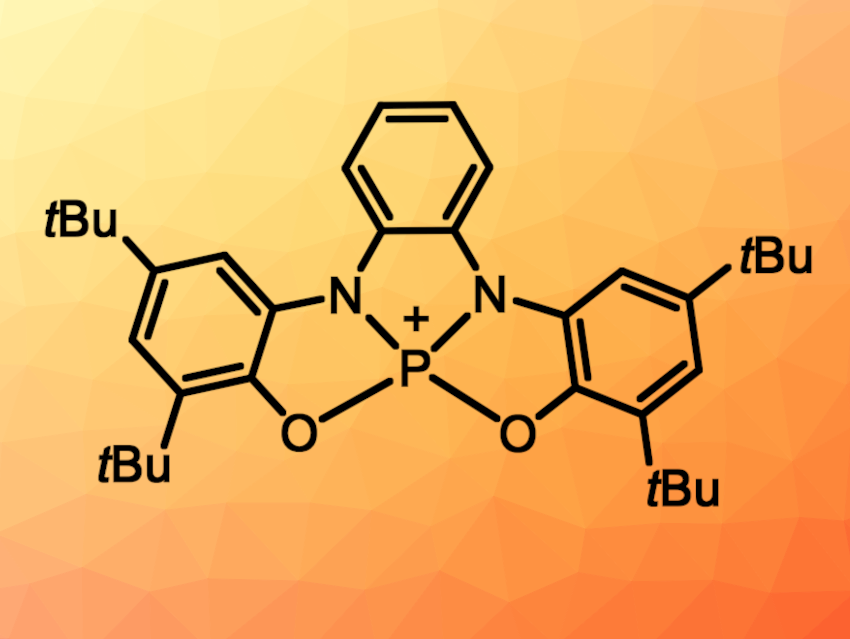
New activation modes in homogeneous catalysis could lead to the development of new catalysts and unprecedented reactions. For example, organocatalysts that are built to show redox cooperativity between a redox-active ligand and the active center—similar to metal-based catalysts—are interesting.
Manuel Alcarazo and colleagues, University of Göttingen, Germany, have used the concept of metal–ligand redox cooperation in the design of an organocatalyst. The team formally replaced a zirconium atom in a known catalyst with a phosphorus atom. The team started from a bis(amidophenolate) ligand, whose protonated form was reacted with PCl3 in the presence of N,N-diisopropylethylamine (DIPEA) to give a phosphorus complex. Treatment of the complex with N-chlorosuccinimide then gave the corresponding chlorophosphorane. A chloride abstraction results in a transient, planar phosphonium cation (pictured above) that can be trapped in the form of its dimethylaminopyridine (DMAP) adduct.
The researchers found that catalytic activity is retained although the phosphonium cation does not contain a metal center. The new complex promotes the catalytic disproportionation of 1,2-(diphenyl)hydrazine into aniline and azobenzene (pictured below).
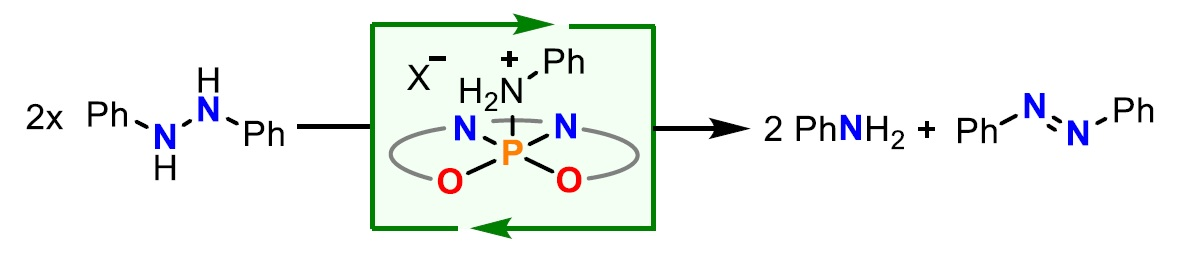
This reactivity implies redox cooperation between the P(V) atom and the ligand. The central P atom acts somewhat like a metal: It offers a coordination position to the substrate and, simultaneously, makes use of the supply of electrons provided by the cooperating ligand. This strategy could offer a new tool for the design of unprecedented redox-active organocatalysts using non-metallic elements such as phosphorus.
- Ligand Enabled Disproportionation of 1,2‐Diphenylhydrazine at a P(V)‐Center,
Manuel Alcarazo, Simon B. H. Karnbrock, Christopher Golz, Ricardo A. Mata,
Angew. Chem. Int. Ed. 2022.
https://doi.org/10.1002/anie.202207450