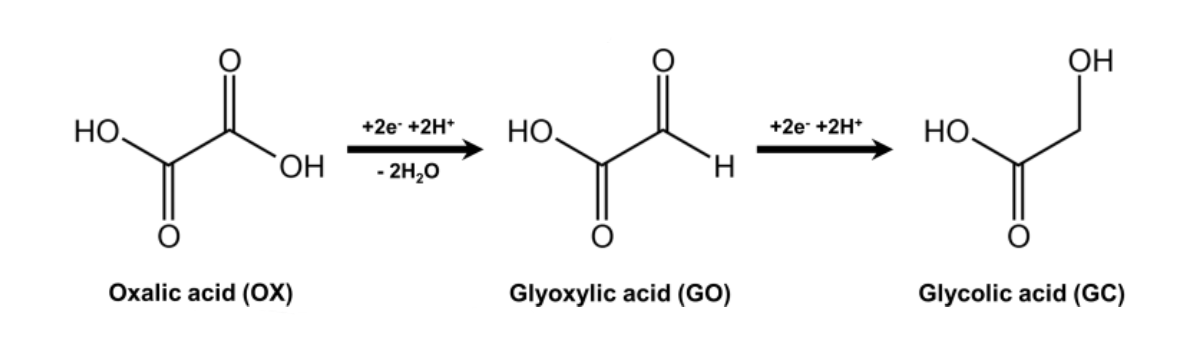
Glycolic acid (GC, pictured below), the simplest hydroxy acid, can be used, e.g., as a feedstock for organic synthesis or as a monomer for biodegradable polymers. It is a value-added chemical with a large potential market. The electrocatalytic reduction of oxalic acid (OX, pictured below) to produce glycolic acid is a green, straightforward process. However, so far, the preparation of glycolic acid via oxalic acid electroreduction generally requires elevated reaction temperatures. The development of processes to efficiently reduce oxalic acid to glycolic acid at ambient temperature would, thus, be useful. Milder conditions also have advantages such as reduced corrosion of the reactor and longer catalyst life.
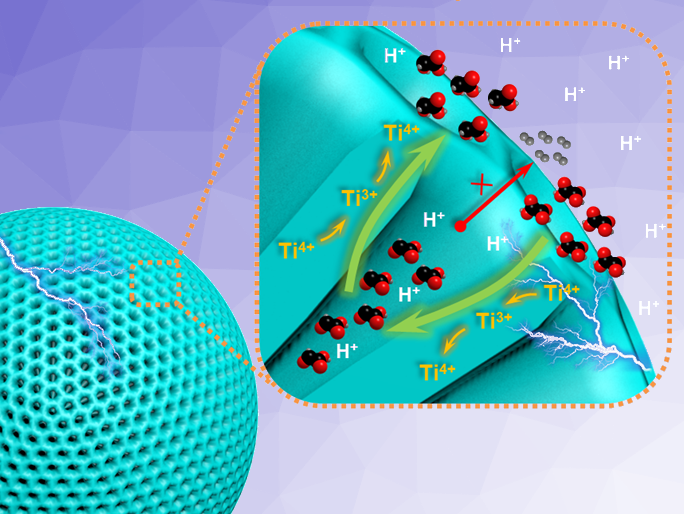
Peng Kang, Tianjin University, China, and colleagues have used mesoporous TiO2 spheres of the anatase phase (schematically pictured at the top) as catalysts to reduce oxalic acid to glycolic acid. The team used NH4OH as a pore-forming agent to obtain the desired TiO2 catalysts using a combination of sol-gel and hydrothermal methods. Pentanoic acid and titanium butoxide were reacted in ethanol, followed by precipitation, the addition of NH4OH, and calcination at 400 °C to give mesoporous TiO2 spheres.
The catalyst showed excellent electrocatalytic activity at ambient temperature, with the Ti3+/Ti4+ redox pair playing an important role. Ti4+ is first reduced to Ti3+, and Ti3+ then serves as the active site for the reduction of oxalic acid. Due to its mesoporous structure, the catalyst has a large specific surface area, exposing more active sites. In addition, the mesoporous structure is critical to allow mild operating conditions. The pores limit the diffusion of the intermediate product glyoxylic acid (GO, pictured above) so that it can be further reduced to glycolic acid. According to the researchers, this work provides a new reference for the electrocatalytic reduction of oxalic acid to glycolic acid on titania-based catalysts with high selectivity.
- Selective Electroreduction of Oxalic Acid to Glycolic Acid by Mesoporous TiO2 Spheres,
Wenjing Xu, Yingying Cheng, Jing Hou, Peng Kang,
ChemCatChem 2023.
https://doi.org/10.1002/cctc.202201687