The anticancer drug cisplatin is often associated with adverse effects (nausea, vomiting, oto- and nephrotoxicity, neuropathy), as well as resistance. Several Ru-based chemotherapeutics show activity against cisplatin-resistant tumors and are advancing in clinical trials due to high delivery into cancer cells and low systemic toxicity.
Sulfur-containing biomolecules are thought to have negative effects on the efficacy of Pt-based anticancer drugs due to irreversible binding of the metal to intracellular thiolato ligands. Several thiol- and sulfur-based nucleophiles have been tested as chemoprotectants to modulate cisplatin nephrotoxicity. For example, sodium diethyldithiocarbamate protects against renal, gastrointestinal, and bone-marrow toxicity induced by cisplatin with no decrease in the antitumor activity. However, protecting normal tissues without inhibiting anticancer activity is challenging.
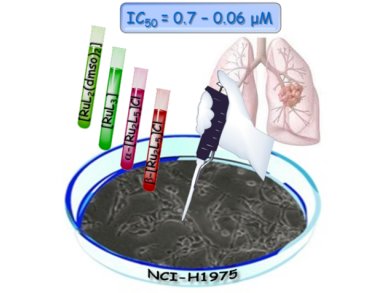
Using these properties, Dolores Fregona and colleagues, University of Padova, Italy, have designed several metal dithiocarbamato complexes that may combine the cytotoxic activity of the metal with the protective effects of a sulfur-based ligand. They now report the synthesis and in vitro cytotoxicity toward non-small-cell lung cancer of RuII and RuIII dithiocarbamato complexes, of which the RuIII complexes show significantly greater activity than cisplatin.
- Ruthenium(II/III)-Based Compounds with Encouraging Antiproliferative Activity against Non-small Cell Lung Cancer,
E. M. Nagy, A. Pettenuzzo, G. Boscutti, L. Marchiò, L. Dalla Via, D. Fregona,
Chem. Eur. J. 2012.
DOI: 10.1002/chem.201202171