Mankind consumes a great deal of tobacco, most commonly in the form of cigarettes. Worldwide, roughly 15,000 kg of nicotine from tobacco makes its way daily into smokers’ lungs. That alone is sufficient reason for us to consider this particular natural product more closely.
In this part we look at what the tobacco plant contains and which chemical changes happen between the tobacco harvest and a finished cigarette.
4. Contents of the Tobacco Plant
Looked at from a chemical standpoint, a tobacco plant consists of precisely those substances that all plants have in common: amino acids, sugars, and fats. However, starting from these primary metabolites, many plants synthesize additional compounds that do not play a direct role in the metabolism of the plant itself, but rather fulfill special ecological functions. Tens of thousands of these so-called “secondary metabolites” are known. Typical examples are fragrances and pigments used to attract insects for pollination purposes, pheromones for attracting partners, or substances to repel or even poison predators.
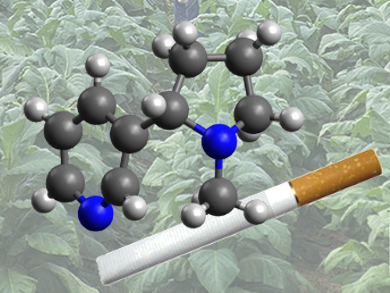
The characteristic secondary metabolite of the tobacco plant, nicotine (1), is synthesized not only by tobacco and other solanaceae species, but also by certain plants completely unrelated to tobacco (see Fig. 1).
|
|
|
Figure 1. The plant toxin nicotine. |
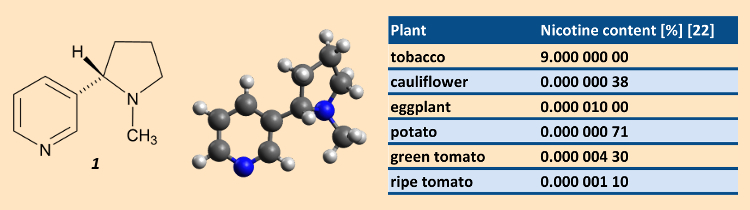
The toxicity with respect to mammals of (S)-(–)-3-(1-methylpyrrolidine-2-yl)-pyridine, nicotine (1), depends heavily upon the species [5]. On the basis of individual cases, the lethal oral dose for adults lies in the range 0.5–1.0 mg/kg body weight, and for infants, ca. 0.1 mg/kg. For humans, nicotine is as toxic as hydrogen cyanide. First mild toxic symptoms include excessive salivation, nausea, stomach pains, vomiting and diarrhea, headache, perspiration, dizziness, and an increase in blood pressure.
The nicotine content of tobacco (dry weight) can be up to 9 %. In comparison, the nicotine content in other plants consumed as vegetables can be regarded as negligible. A typical cigarette tobacco contains ca. 1.5 % (S)-(–)-nicotine, which at over 95% is the principal alkaloid present [6]. There are also small amounts of the enantiomeric (R)-(+)-nicotine (2) [7] together with nornicotine (3), anatabine (4), and anabasine (5) [8] (Fig. 2). In the course of drying and fermentation, some of the alkaloid content of tobacco leaves is decomposed, leading to compounds (6) – (14) [9].
|
|
|
Figure 2. Nicotine and related tobacco alkaloids. |
5. From Tobacco Plant to Cigarettes
Today’s market for tobacco products is dominated (96 %) by cigarettes. Most of the tobacco varieties utilized for this purpose are bred from the species Nicotiana tabacum, known as Virginia tobacco. Only mahorka tobacco, employed chiefly in Russia, is derived instead from Nicotiana rustica.
The tobacco plant stemmed originally from South America, but it has long been cultivated throughout the world. The principal alkaloid in tobacco is nicotine, a colorless liquid when isolated. Tobacco thrives especially in warm climates, although it is even cultivated in Germany. German tobacco lacks a particularly good flavor, contains little nicotine, and generally is used only in mixtures with other tobaccos.
The annual world production of tobacco is roughly 7 million metric tons, from a total of 3.8 million hectares of cultivated land. In 2011, the largest sources of tobacco, apart from China, were India, Brazil, the USA, Argentina, and Malawi. Countries showing the greatest growth in tobacco production were all in Africa: Mozambique, Zambia, Mali, and Ghana [10].
Tobacco cultivation often entails utilization of large amounts of nitrate-containing fertilizers. These result not only in enhanced plant growth, but also certain side effects that in the end are of considerable significance in tobacco consumption. Increased uptake of nitrates from the fertilizer makes it possible for the plants to synthesize more nicotine as they grow, but also increases the nitrate content in the plants themselves, which in the smoking process leads to a higher level of carcinogenic substances in the smoke [11]. After the plant leaves have been harvested, the processes of drying and especially of fermentation lead to further major changes in leaf composition.
|
From the Plantation to the Cigarette pack |
|
|
1 Tobacco Three tobacco varieties dominate from a cultivation standpoint: virginia, oriental, and burley. The tiny tobacco seeds (a gram contains 12,000!) are mixed with water and poured into the soil. Tobacco plants grow up to 2 m high, and require two to three months before they are ready for harvesting. |
2 Harvesting In the case of virginia and oriental tobaccos, individual leaves are harvested, whereas with burley, entire plants can be utilized. Some tobacco harvesting can be conducted mechanically, but most of the work is still done by hand. |
|
3 Drying All tobacco is threaded, leaf by leaf, on long cords and hung up for drying. The large leaves of virginia tobacco are dried over the course of only 4–7 days in streams of hot air. Leaves of oriental tobacco are dried in the sun, which requires about four weeks, whereas the dark brown burley tobacco hangs for 6–8 weeks in a well-ventilated barn. |
4 Fermentation Fermentation is the most time-consuming step. The tobacco is allowed to “rest” in large stacks for as long as several months. During this time it is important that the stacks be turned every now and then so that all the leaves ferment uniformly. During this fermentation proess, temperatures of 50–60 °C are attained, in the course of which proteins and some of the nicotine are degraded, and countless new substances arise which confer its typical aroma upon the tobacco. |
|
5 Choosing In the course of purchasing tobacco, the processing industry tests the quality of the product on offer with respect to aroma, flavor, and optical impact; then warehousing and other logistics must be arranged, because tobacco always needs to be “rested”. During the storage period, tobacco develops its full aroma. |
6 Mixing Each cigarette variety involves the mixing of 30 to 50 portions of tobacco. Mainly, the somewhat sweet oriental, the aromatic virginia, and the more “spicy” burley are utilized. Experts ensure consistency in the taste composition of individual blends. |
|
7 Seasoning and Cutting Dried crude tobacco is moistened with steam and sprayed with flavoring agents. Every brand has its own unique recipe. The tobacco is now cut mechanically into fibers exactly 0.75 mm wide. |
8 Finishing A 4–6 km long paper strip (sufficient for 15 minutes of production time!) is imprinted with trademarks, provided with a track of glue, and charged with the prerolled tobacco. Next it is rolled, glued, cut, and wrapped, as appropriate, with a filter, including the requisite mouthpiece paper. |
6. A Tiny Chemical Factory, Functional Even After the Harvest
“Fermentation” refers to modification and degradation processes brought about largely by enzymes in the plants themselves, or by microbial influences. These contribute to tobacco’s typical aroma and dark color; the connoisseur would say they make it “smokable”. Depending upon the conditions selected – including temperature, moisture, and compression of the tobacco – various degradation processes occur in the leaves, which lead to sundry reaction products. Taken as a whole, and from a chemical standpoint, the fermentation process remains rather unclear, and processing steps employed are based above all on long practical experience. Nevertheless, the chemist can recognize a few fundamental aspects.
Essentially, during fermentation carbon dioxide forms, heat is released, and the pH falls [12]. Perhaps surprisingly, the nitrogen content of the tobacco leaf changes very little, because proteins are degraded to smaller nitrogen-containing compounds with very little overall nitrogen loss. The aroma of the smoke is significantly improved, since combustion of proteins causes formation of components lending unpleasant aromas. The nicotine content decreases by 30–50 % during drying and fermentation, and many degradation products can be established in fermented tobacco (Fig. 2). Regardless of the particular reaction cascades that take place during tobacco leaf fermentation, however, the tobacco tastes better afterward.
Cigarette tobaccos are always mixtures of different varieties. In processing, it is normally only leaves that are utilized. The leaf-veins are first removed, although these are often chopped up and later put back into the tobacco mixture. Mixing can be used to ensure that the nicotine content falls below some legally established limit, e.g., 1 mg in the smoke from a single cigarette [13].
The third part of this series.
References
[5] ChemIDplus Lite.
[6] N. L. Benowitz et al., Handb. Exp. Pharmacol. 2009, 192, 29. DOI: 10.1007/978-3-540-69248-5_2
[7] D. W. Armstrong et al., Chirality 1998, 10, 587. DOI: 10.1002/(SICI)1520-636X(1998)10:7<587::AID-CHIR6>3.0.CO;2-#
[8] P. Jacob III et al., Am. J. Publ. Health 1999, 89, 731. DOI: 10.2105/AJPH.89.5.731
[9] E. Leete, Science 1965, 147, 1000. DOI: 10.1126/science.147.3661.1000
[10] http://www.tobaccoatlas.org/industry/growing_tobacco/text.
[11] M. Pötschke-Langer et al. in Alkohol und Tabak (Eds.: M. Singer, A. Batra, K. Mann), Thieme Verlag, Stuttgart (in German), 2011, 91. ISBN: 978-3-13-146671-6
[12] C. Pyriki, Z. Lebensm. Unters. Forsch 1951, 92, 322 (in German). DOI: 10.1007/BF01088858
[13] Deutsches Krebsforschungszentrum (Eds.), Tabakatlas Deutschland, Steinkopff-Verlag Darmstadt (in German), 2009. ISBN: 978-3798518827
The article has been published in German as:
- Starker Tobak – Unsere Lust und Last mit der Zigarette,
Sabine Streller, Klaus Roth,
Chem. Unserer Zeit 2013, 47, 248–268.
DOI: 10.1002/ciuz.201300636
and was translated by W. E. Russey.
The Chemistry of Tobacco – Part 1
Looking at the history of tobacco consumption – from chewing and snuffing to smoking
The Chemistry of Tobacco – Part 2
What does tobacco contain and which chemical changes happen between the harvest and a finished cigarette?
The Chemistry of Tobacco – Part 3
How does a tobacco plant synthesize nicotine?
The Chemistry of Tobacco – Part 4
What does cigarette smoke contain and what does nicotine do to the smoker?
The Chemistry of Tobacco – Part 5
What happens when a smoker tries to quit?
See all articles by Klaus Roth published by ChemistryViews magazine
.jpg)



