After carrying out the “Three-Step Preparation of Espresso,” (see part 1) the thrill of anticipation commences with a scrutinizing glance at the crema, the foamy, consolidated surface layer of an espresso. This arises through an interplay of emulsified fats, denatured proteins, and surface-active components.
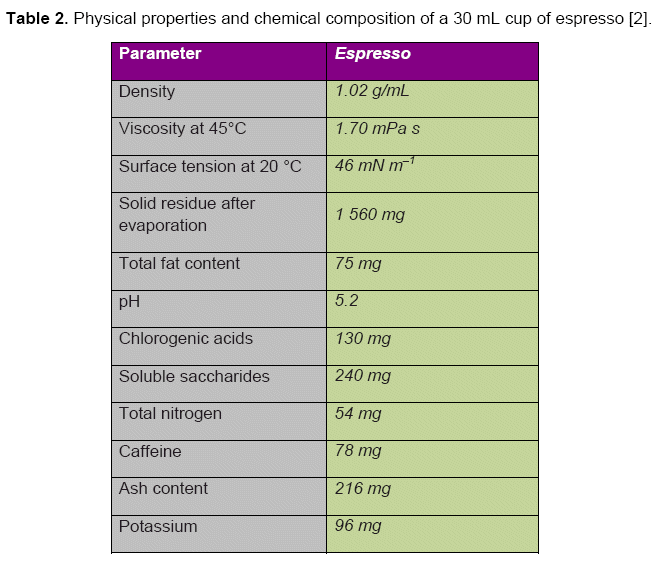
The surface tension in espresso is only half that of pure water (see Tab. 2). The structures of the surfactants responsible are unknown; they may be products from reactions between saccharides and proteins (glycoproteins) or lipids (glycolipids).
The texture of the crema is a mark of quality: it must be stable, medium brown, and pervaded by wispy, lighter stripes (“tiger-skin effect”). Even after vigorous stirring the crema must again consolidate itself. A light, thin, and relatively unstable crema points to insufficient extraction, a consequence of too coarse a grind or too low a water temperature (see Fig. 4). If the crema is dark brown with a hole in the middle, the coffee powder was insufficiently porous, or too much coffee was used.
A consolidated crema retains volatile aromatic substances, prevents the espresso from cooling too rapidly, and protects against hydrolysis of aromatic substances dissolved in fats.

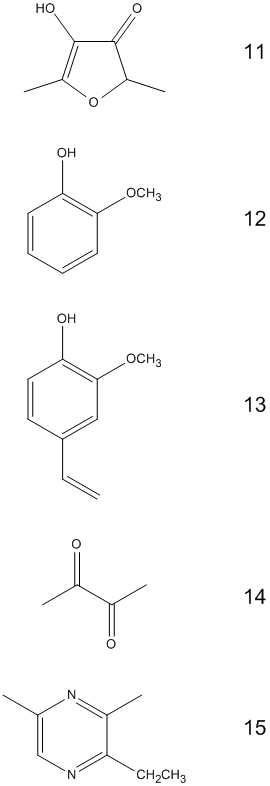
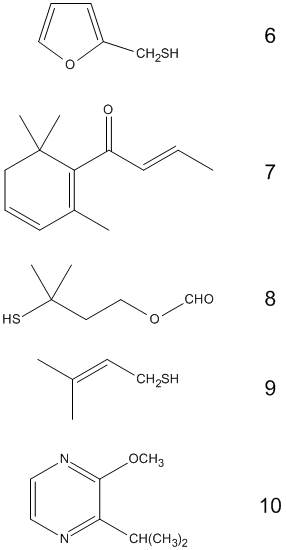
Before we’re able to take our first sip, the coffee aroma, with its 800 so far identified components, will already have stroked the 30 million olfactory receptor cells in our two nostrils. These receptor cells direct their excitation along the shortest possible path to the cerebral cortex for processing. Many of the compounds in coffee fragrance are present in other roast aromas as well (malt, grilled meat) [10]. The structural diversity of the aromatic components of coffee could fill an organic chemistry textbook [11]. Many of the compounds are present only at a trace level, however, below the olfactory threshold, and presumably play no significant part in the aroma. The coffee fragrance “top ten” are collected in Tab. 3.
The crema brings another benefit as well: its body. This refers to the creamy consistency that one perceives through the tongue and gums. “Body” in this sense as it relates to a liquid is readily illustrated by drinking milk and then water. Even with eyes closed and the nose firmly held one senses the difference between the two, namely through the body of the respective fluid.
The flow characteristics associated with espresso are quite unique. On one hand the lipids and dissolved polysaccharides [12] lead to a three-fold increase in viscosity: from 0.61 for pure water to ca. 1.7 mPa•s. On the other hand, surfactant components reduce the surface tension from 73 to 46 mN•m–1, so that gum and tongue surfaces are moistened more readily.
After the first sip, the mouth area is coated with crema, which has two important consequences:
- the receptors for bitterness, located toward the back of the tongue, are partially covered, so the subjective sense of bitterness is somewhat attenuated, and
- because the crema adheres firmly to the mouth area, aromatic and flavor components dissolved in it are released only gradually.
After enjoying a cup of espresso with a good crema, one can actually still smell and taste it 20 – 30 min later.
All Four Flavors
Espresso addresses sensory cells in the 3000 taste buds on the surface of the tongue with respect to all four taste qualities: sour, salty, sweet, and bitter.
Sour
Like every coffee, espresso is slightly acidic. The reduction in pH is caused by phosphoric acid and over 60 different organic acids, with acetic acid and citric acid present to the greatest extent. A light acidity is an indicator of quality, however, because coffee in the absence of acid tastes flat and bitter.
A distinction must be made between this and the slowly declining pH that results from allowing hot coffee to stand. In the latter case esters are cleaved, and the resulting free acids are what lower the pH further. This provides a rational explanation for the unwholesomeness of the coffee that, in many offices, stands around on hotplates for hours on end. Such of course cannot occur with espresso itself, which is always prepared to order; i.e., one always waits for the espresso, never the reverse!
Sweet
It is the soluble saccharides in espresso that lead to the coveted slight sweet tone.
Salty
In the course of extraction, salts present in finely ground coffee are largely eluted. About 40 % are potassium salts, together with salts of magnesium, iron, and copper. These traces of salt strengthen espresso’s taste in a positive sense.
The flavor of ordinary filter coffee can also be improved by addition of small amounts of table salt.
Bitter
The bitter taste of espresso has a number of sources. Caffeine, various phenols, and trigonelline that has not decomposed during the roasting process all contribute to bitterness. Most of the not yet characterized bitter components arise during roasting. Roasting for a very short period of time (< 2 min) with very hot gas (300 – 400 °C) results in beans that are more bitter.
The question of whether bitterness is perceived to be an unpleasant characteristic depends upon the culture:
- In the United States and Northern Europe, espresso is brewed almost exclusively from high-quality Arabica beans.
- Around the Mediterranean, Arabica is blended with a certain amount of Robusta, a mixture with a more bitter flavor.
In northern latitudes an attempt is often made to prepare espresso that is not as strong (=bitter) by using somewhat more than 50 mL of water. The effort is doomed to failure, however, since overextraction produces an unpleasant woody and astringent tone (often mistaken for bitterness).
Other Senses
There is one other sense we should not forget: the sense of hearing. Since a good espresso is served in prewarmed cups, and drunk immediately thereafter, it is relatively hot, and drawn into the mouth with a soft slurping sound, intended only for one’s own ear. This slurping is, in reality, a steam distillation of the espresso, through which less volatile aromatic components are condensed along with steam in the entire respiratory tract, and then gradually exhaled and thereby sensed yet again in the nasal passages.
Physiological Effect
Caffeine, 1,3,7-trimethylxanthine, is the physiologically active constituent in coffee. A cup of espresso contains ca. 60–70 mg, a cup of brewed coffee 100–150 mg, and a glass of cola 40–60 mg of caffeine.
Caffeine’s stimulative effect accounts for the great popularity of coffee, which can almost verge on reverence. The assertion of the Scottish philosopher, James McKintosh, that “the powers of a man’s mind are proportionate to the quantity of coffee he drinks” is to be doubted, however, although many reknowned intellectuals such as Johann Sebastian Bach, Balzac, Kant, Hemingway, Baudelaire, Voltaire, Thomas Mann, etc. also insisted that only with coffee were they able to complete their masterpieces.
Caffeine exerts its physiological effects by intervening in the regulatory mechanism of neuron activity [13]. Long-term neuronal activity during the waking phase leads to an increase in the local adenosine concentration. Special receptors bind this adenosine, neuron activity is attenuated, and one becomes tired.
This is a classic feedback loop, through which the reaction product adenosine retards its own production [14] – and as a result of which, within certain limits, the adenosine concentration remains constant. Caffeine can bind to the same receptors, and block them against attack from adenosine, so that the retardation effect is eliminated, weariness disappears, and one remains in an alert condition. Caffeine thus acts indirectly as a stimulant by switching off the damping effect of adenosine.
Large amounts of caffeine have little effect, however: It is known from animal studies that intake of amounts of caffeine equivalent to one to four cups of strong coffee in humans produces the desired increase in activity [15]; but with higher doses the effect of the caffeine is reversed: after ingestion of the human equivalent of ten cups of coffee, mice become calmer and more tired than in the absence of caffeine.
Acknowledgement
I wish to thank Professor W. Tressl, Berlin, for his helpful support.
References
10. R. Tressl, D. Rewicki in Flavor Chemistry. Thirty Years of Progress (Eds: R. Teranishi, E. L. Wick, I. Hornstein), Kluwer/Plenum, 1999, 305.
11. W. Gosch, Chem. unserer Zeit 1996, 30, 126. DOI: 10.1002/ciuz.19960300304
12. M. Coimbra, F. Nunes, J. Agric. Food Chem. 1997, 45, 3238. DOI: 10.1021/jf970009t
13. Intelligible description of the physiological effects of coffee: S. Braun, Buzz – The Science and Lore of Alcohol and Caffeine, Oxford University Press, Oxford, 1996.
14. D. G. Rainnie, H. C. R. Grunze, R. W. McCarley, R. W. Greene, Science 1994, 263, 689. DOI: 10.1126/science.8303279
15. It was recently shown that coffee intake also leads to an increase in the adenosine level in plasma: L. A. Conlay, J. A.Conant, F. de Bros, R. Wurtmann, Nature 1997, 389, 136. DOI: 10.1038/38160
Author
Professor Klaus Roth, Freie Universität Berlin, Germany.
The article has been published in German in:
and was translated by E. Russey.
Other articles by Klaus Roth published by ChemistryViews magazine:
- In Sparkling Wine, Champagne & Co
Klaus Roth shows that only chemistry can be this tingling
DOI: 10.1002/chemv.201000042 - In Chemistry of a Hangover — Alcohol and its Consequences
Klaus Roth asks how a tiny molecule like ethanol can be at the root of so much human misery?
DOI: 10.1002/chemv.201000074 - In Chocolate — The Noblest Polymorphism
Klaus Roth proves only chemistry is able to produce such a celestial pleasure
DOI: 10.1002/chemv.201000021 - In The Chemist’s Fear of the Fugu
Klaus Roth shows the chemist’s fear of the fugu or pufferfish extends as far as the distinctive and intriguing poision it carries
DOI: 10.1002/chemv.201000104
Also of Interest