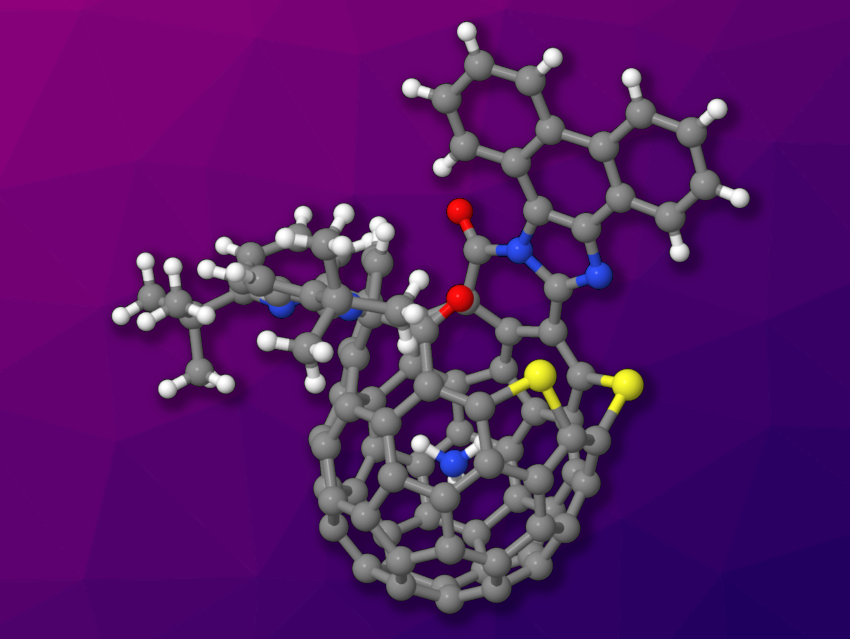
Molecules that combine different carbon allotropes, such as hybrids of fullerenes and graphenes or fullerenes and carbon nanotubes, can have interesting and useful properties. However, such species can be difficult to synthesize and characterize.
Yasujiro Murata, Kyoto University, Japan, and colleagues have developed a method for the synthesis of π-extended fullerenes, which turned out to encapsulate in-situ generated NH3 (example pictured). The team started from an open form of a C60 fullerene that is functionalized with a 1,2-dicarbonyl unit. This fullerene was reacted with diamines to extend the molecule’s skeleton. The team used 4,5-dimethyl-1,2-phenylenediamine, 2,3-diaminonaphthalene, or 9,10-diaminophenanthrene. This created fullerenes with fused pyrazine or imidazole units. The condensation reactions form ammonia, which is encapsulated in the fullerene.
The combination of the planar π-systems with the curved π-system of the C60 fullerene resulted in an increase of absorption coefficients in the visible region. In addition, the researchers found that one system with an encapsulated NH3 molecule undergoes a sustained release of ammonia. This process causes an expansion of the opening over time, leading to a large opening with a ring-atom count of 19. According to the team, this type of timed reaction could provide a way to introduce additional functions in related fullerene–graphene hybrid molecules.
- π-Extended Fullerenes with a Reactant Inside,
Yoshifumi Hashikawa, Nana Fujikawa, Yasujiro Murata,
J. Am. Chem. Soc. 2022.
https://doi.org/10.1021/jacs.2c12259