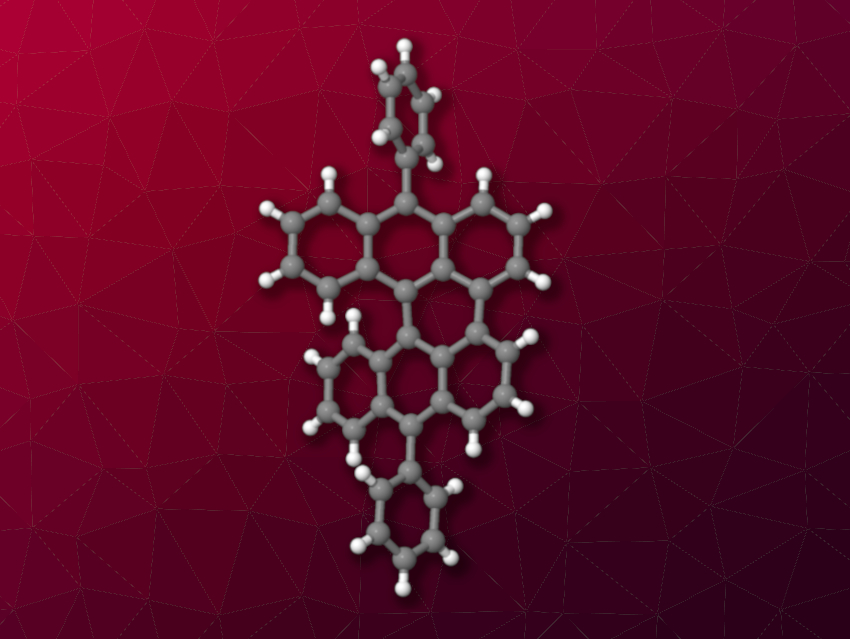
Chemical actinometers are systems that can be used to measure a photon flux. They are useful for measuring the incoming light energy in a photochemical reaction and determining the quantum yield. Mesodiphenylhelianthrene (MDH, pictured), for example, can be used as a chemical actinometer. Under light irradiation, it undergoes self-oxidation into an endoperoxide. This reaction has a known quantum yield and can be followed using spectroscopic methods to measure a photon flux. However, MDH is not commercially available, and the structure of the endoperoxide had not been confirmed so far.
Bernhard Spingler, University of Zurich, Switzerland, and colleagues have developed a five-step synthesis of MDH from a commercially available compound and characterized the endoperoxide. The team started from 1-aminoanthraquinone, which was converted to 1-chloroanthraquinone via reactions with tert-butyl nitrite and CuCl2. The resulting intermediate was converted to 1,1′-dianthraquinone using an Ullmann reaction. A Cu-catalyzed ketone-ketone reductive coupling was used to close the final six-ring in the core of MDH. The remaining two ketone groups were reacted with phenyl lithium to introduce the phenyl substituents, followed by dehydration to give MDH.
Single-crystal X-ray diffraction showed that MDH has a slightly twisted structure (pictured). It is light-sensitive, and a violet solution of MDH turns yellow under visible light irradiation due to the formation of the corresponding endoperoxide. The researchers used density functional theory (DFT) calculations and 2D NMR spectroscopy to elucidate the structure of the endoperoxide. They found evidence for the formation of the peroxide across a ring next to one of MDH’s phenyl substituents.
- Synthesis of Mesodiphenylhelianthrene from 1-Aminoanthraquinone and the Structural Elucidation of Its Endoperoxide Species after Irradiation,
Sheng-Jan Feng, Frank Schumer, Eva Vandaele, Giuseppe Meola, Flavia Kradolfer, Sandra Luber, Bernhard Spingler,
Org. Lett. 2022.
https://doi.org/10.1021/acs.orglett.2c01758