Tungsten and molybdenum iodide clusters can have interesting photophysical properties, including a bright red phosphorescence and/or singlet-oxygen generation. They could have applications in various fields, e.g. in photocatalysis, tumor treatment, or the photodynamic inactivation of bacteria. Compared with anionic cluster species of this type, investigations regarding the luminescence of neutral and cationic clusters are scarce.
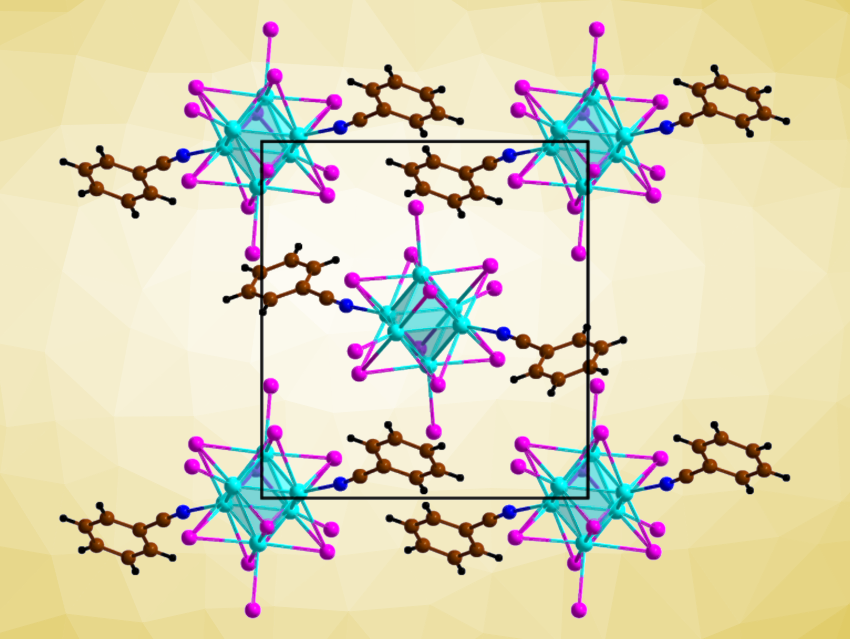
Hans-Jürgen Meyer, University of Tübingen, Germany, and colleagues have synthesized a previously unknown, photoactive neutral tungsten iodide cluster, i.e., [W6I12(NCC6H5)2] (crystal structure pictured). The cluster was obtained via a reaction of Cs2W6I14, ZnI2, and benzonitrile under solvothermal conditions at 200 °C. It was obtained in the form of orange cube-shaped crystals with a red-to-orange photoluminescence, together with Cs2ZnI4 as a byproduct.
The crystal structure shows a dense packing of molecular cluster units, interconnected by a network of hydrogen bonds. This structure results in a high thermal stability, with decomposition temperatures above 400 °C. Usually, ligand-substituted tungsten or molybdenum halide clusters show low stability against water, but [W6I12(NCC6H5)2] showed no sign of hydrolysis even after being suspended in water for over a month. The researchers also investigated the cluster’s photocatalytic properties. They found that it can successfully act as a photocatalyst in the decomposition of rhodamine B in water.
- The Remarkably Robust, Photoactive Tungsten Iodide Cluster [W6I12(NCC6H5)2],
Florian Pachel, Markus Ströbele, Carl P. Romao, David Enseling, Thomas Jüstel, Hans Jürgen Meyer,
Eur. J. Inorg. Chem. 2023.
https://doi.org/10.1002/ejic.202300096