The introduction of fluorine atoms into organic compounds can be used to tune their properties, such as lipophilicity, solubility, and metabolic stability. This can be particularly useful in pharmaceutical chemistry or agrochemistry. Often, trifluoromethyl groups are used in this context. N-CF3-functionalized amines, for example, can be prepared using electrophilic trifluoromethylation reagents. N,N-bis(trifluoromethyl)amines with two CF3 groups show improved lipophilicity and stability, but are generally more challenging to prepare. Similar structures could provide an easier-to-access alternative.
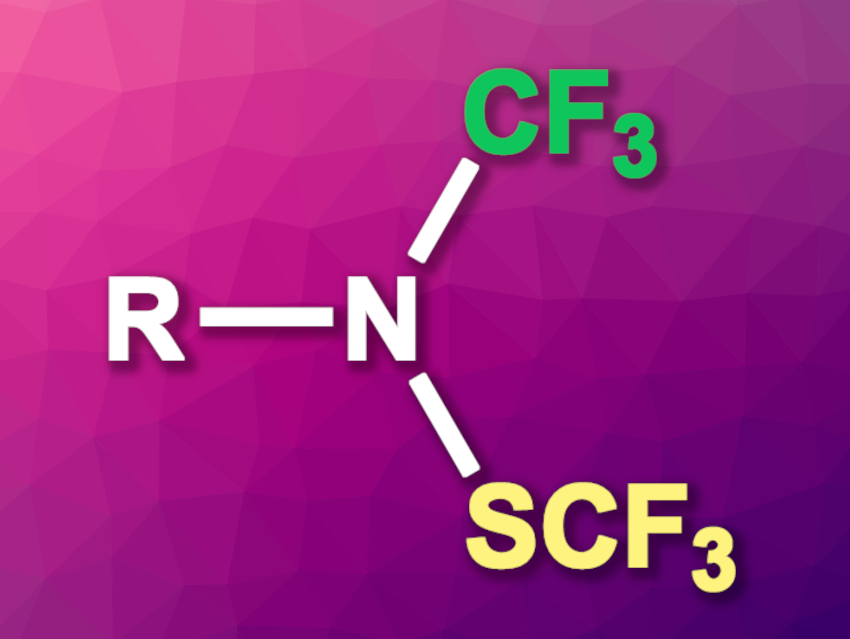
Anis Tlili, Université Lyon 1, Centre national de la recherche scientifique (CNRS), École supérieure de chimie, physique, électronique de Lyon (CPE-Lyon), Institut National des sciences appliquées (INSA), Villeurbanne, France, and colleagues have developed mild and efficient protocols for accessing novel N-((trifluoromethyl)thio)-N-(trifluoromethylamines), i.e., compounds of the type RN(SCF3)(CF3) (pictured).
The team started from isothiocyanates (R–NCS), which were reacted with N-trifluoromethylthiophthalimide as an electrophilic trifluoromethylthiolation reagent in MeCN at room temperature in the presence of CsF. Under these conditions, different benzylic, aliphatic, and aromatic isothiocyanates were converted to the desired products in moderate to good yields.
Aromatic substrates with strong electron-withdrawing groups did not react under these conditions. For these substrates, the team modified their approach: Using a two-chamber reactor to avoid mixing all components, they reacted an isothiocyanate and AgF in one chamber. The Langlois reagent (sodium trifluoromethanesulfinate, CF3SO2Na) was reacted with chloro diphenylphosphine (Ph2PCl) in the second chamber to create the trifluoromethylthio dimer (SCF3)2, which is transferred to the first chamber. The researchers demonstrated the scalability of this protocol by performing a gram-scale reaction.
- Novel N(SCF3)(CF3)-Amines: Synthesis, Scalability and Stability,
Yi Yang, Nathalie Saffon-Merceron, Julien C. Vantourout, Anis Tlili,
Chem. Sci. 2023.
https://doi.org/10.1039/d2sc06542h