Binary crystalline nitrides of group 15 elements have not been well-explored in chemical research so far. This is due to their challenging synthesis, requiring harsh pressure and temperature conditions, and so far, examples have been limited to phosphorus and arsenic nitrides. Thus, antimony is the next target for the discovery of crystalline pnictogen nitrides.
Matteo Ceppatelli, LENS, European Laboratory for Non-Linear Spectroscopy, Firenze, Italy, and ICCOM-CNR, Institute of Chemistry of Organometallic Compounds, National Research Council of Italy, Firenze, and colleagues have synthesized crystalline Sb3N5 via a high-pressure and high-temperature chemical reaction of antimony and nitrogen in a laser-heated diamond anvil cell. A reaction was observed when compressing Sb in the presence of N2 at 32–35 GPa and laser heating at this pressure to temperatures of 1600–2200 K. These conditions correspond to the fluid phases of Sb and N2. The reaction was observed using single-crystal synchrotron X-ray diffraction (XRD). The diffraction patterns acquired after the reaction at the center of the laser-heated area show the complete consumption of crystalline Sb and the formation of a new crystalline product.
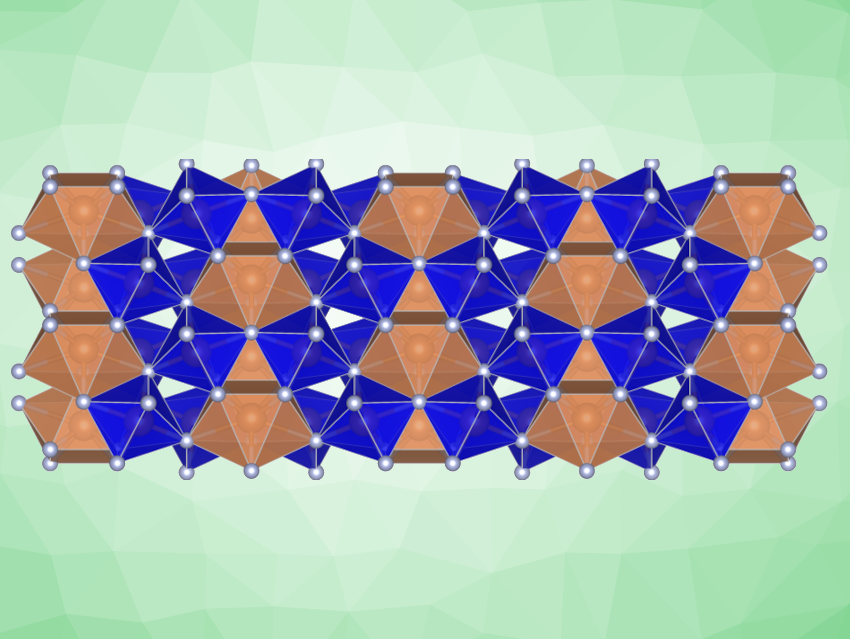
Using XRD, the team solved the structure of the product. They found that Sb3N5 has a structure belonging to the orthorhombic space group Cmc21. It contains two types of Sb atoms in distorted octahedral and trigonal prismatic/square antiprismatic coordination, respectively. They form bilayers of octahedra and monolayers of square antiprisms, respectively. Only chemical bonds between Sb and N are present, indicating the complete cleavage of the N≡N triple bond. The crystal structure of Sb3N5 persisted at the pressure of synthesis without decomposition, indicating Sb3N5 could be stable or metastable at about 35 GPa and room temperature.
- High‐pressure and high‐temperature synthesis of crystalline Sb3N5,
Matteo Ceppatelli, Manuel Serrano-Ruiz, Marta Morana, Kamil Dziubek, Demetrio Scelta, Gaston Garbarino, Tomasz Poręba, Mohamed Mezouar, Roberto Bini, Maurizio Peruzzini,
Angew. Chem. Int. Ed. 2023.
https://doi.org/10.1002/anie.202319278