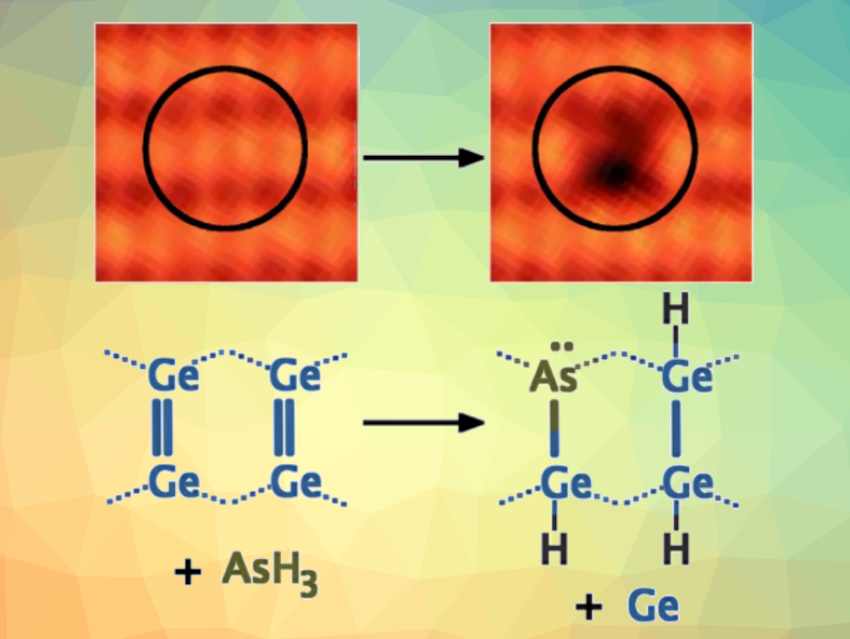
Germanium is a promising material for spintronics (electronics that depend not only on charge, but also on electron spin) and quantum information applications, with significant advantages over silicon. However, research efforts aiming to create atomic-scale devices such as qubits using donor atoms have largely focused on phosphorus in silicon. The low success rate of the thermal annealing step required to incorporate phosphorus atoms into silicon crystals has created doubts that this method can be scaled up to fabricate large numbers of qubits.
Steven R. Schofield, University College London, UK, and colleagues have found that arsenic atoms can be fully incorporated into the (001) surface of germanium at room temperature (pictured). The team exposed atomically clean germanium (001) surfaces to arsine (AsH3) under ultra-high vacuum conditions and observed changes using scanning tunneling microscopy (STM). They used density functional theory (DFT) calculations to model the surface and interpret the observed surface features and dynamics.
Since no thermal anneal is required for the incorporation of arsenic into germanium, the donor incorporation probability is significantly improved compared with phosphorus in silicon. Compared with silicon, germanium has higher electron mobility and stronger spin-orbit coupling. According to the researchers, the work could be helpful for the scaled-up fabrication of donor-based quantum technological devices.
- Room Temperature Incorporation of Arsenic Atoms into the Germanium (001) Surface,
Emily V. S. Hofmann, Taylor J. Z. Stock, Oliver Warschkow, Rebecca Conybeare, Neil J. Curson, Steven Scholfield,
Angew. Chem. Int. Ed. 2022.
https://doi.org/10.1002/anie.202213982