Teflic acid, or pentafluoroorthotelluric acid, is a strong acid with the formula HOTeF5. It is a fluorinated derivative of orthotelluric acid (Te(OH)6). The teflate, or pentafluoroorthotellurate, group (OTeF5) is stable against oxidization and electrophilic attack and has strong electron-withdrawing properties. It can, thus, be used, e.g., in weakly coordinating anions or strong Lewis acids. However, the group is sensitive to hydrolysis and can form HF as a degradation product. This means compounds with a teflate group usually require inert conditions, which hampers their use.
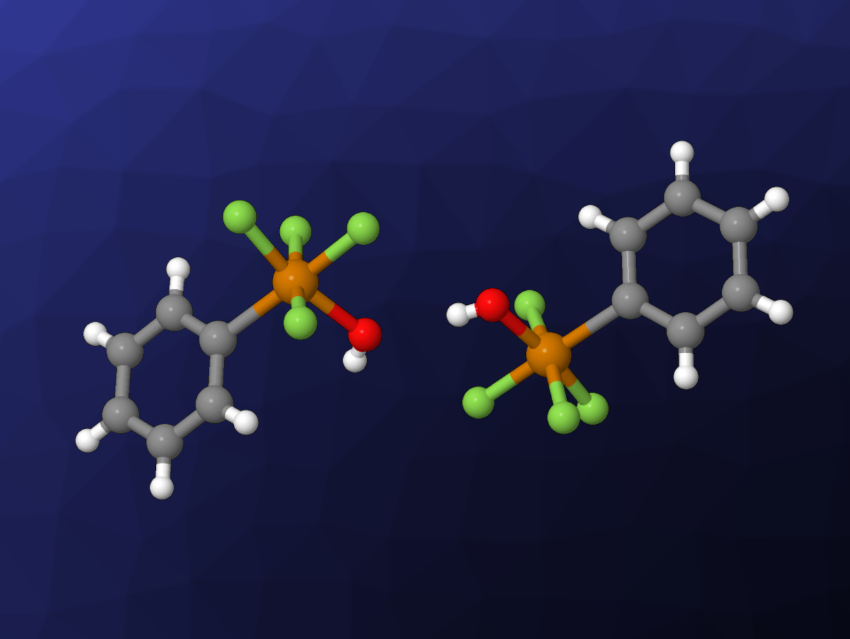
Sebastian Riedel, Free University Berlin, Germany, and colleagues have synthesized air-stable derivatives of pentafluoroorthotellurate substituted with fluorinated or nonfluorinated aryl groups. The team first prepared the acid cis-PhTeF4OH (pictured) from PhTeF5 bis dissolving it in a MeCN/water mixture. According to the researchers, this product is the first crystallized species with a TeF4OH moiety connected to an aryl group. It was further transformed to Ag[cis-PhTeF4O] in two steps: a reaction with Me3SiCl to give cis-PhTeF4OSiMe3, followed by a reaction with AgF. The silver salt was then used as a cis-PhTeF4O transfer reagent, e.g., in a reaction with [PPh4]Cl that gives the salt [PPh4][cis-PhTeF4O].
The team also prepared derivatives with fluorinated aryl groups. They obtained trans-(C6F5)2TeF4 via a fluorination of Te(C6F5)2 using trichloroisocyanuric acid, potassium fluoride, and trifluoroacetic acid. Further transformations gave K[trans-(C6F5)2TeF3O] and trans-(C6F5)2TeF3OH. K[trans-(C6F5)2TeF3O] is both moisture- and air-stable, as well as soluble in organic solvents. Overall, the work could be useful for the synthesis of new weakly coordinating anions or Lewis acids.
- Air-stable Aryl Derivatives of the Pentafluoroorthotellurate,
Daniel Wegener, Kurt F. Hoffmann, Alberto Pérez-Bitrián, Ilayda Bayindir, Amiera Nadien Hadi, Anja Wiesner, Sebastian Riedel,
Chem. Commun. 2022.
https://doi.org/10.1039/d2cc03936b