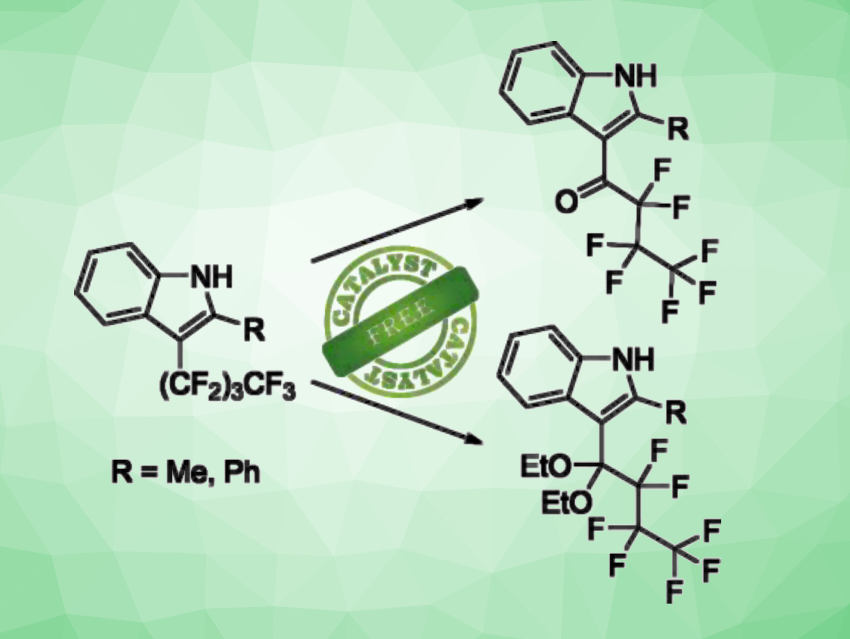
Due to their unique properties, fluorinated organic compounds are useful, e.g., in pharmaceutical chemistry or materials chemistry. Hence, there is great interest in developing new synthetic routes to introduce fluorine into organic compounds or to modify fluorinated substructures, which can be challenging due to the high C−F bond strength.
Ulrich Tallarek, University of Marburg, Germany, and colleagues have developed a catalyst-free method that allows the indirect selective C–F activation of a perfluorinated side chain in the C(3) position of 1H-indoles under mild reaction conditions (pictured). The team found that in the presence of water and diazabicycloundecene (DBU) as a base, 2-methyl-3-(perfluorobutyl)-1H-indole is hydrolyzed to give a ketone derivative (pictured in the top right corner). When sodium ethoxide is used as a nucleophile instead of water, a ketal can be obtained (pictured in the bottom right corner).
The electron-withdrawing effect of the perfluorinated alkyl group allows the deprotonation of the amino function of the indole using a base. This generates an alkene, which enables a nucleophile to be inserted in the α-position of the perfluorinated side chain of indoles.
The method provides a pathway for the generation of heptafluoro-1-indol-butan-1-ones using inexpensive reagents. It provides access to building blocks for the synthesis of biologically active molecules. 3-Substituted 2-phenylindoles, in particular, are promising structures for medicinal chemistry.
- Catalyst‐Free Indirect C–F Activation of 3‐Perfluoroalkyl Indoles,
Thomas Roider, Olaf A. Kleykamp, Sergei I. Ivlev, Reinhard W. Hoffmann, Ulrich Tallarek,
Eur. J. Org. Chem. 2022.
https://doi.org/10.1002/ejoc.202201025