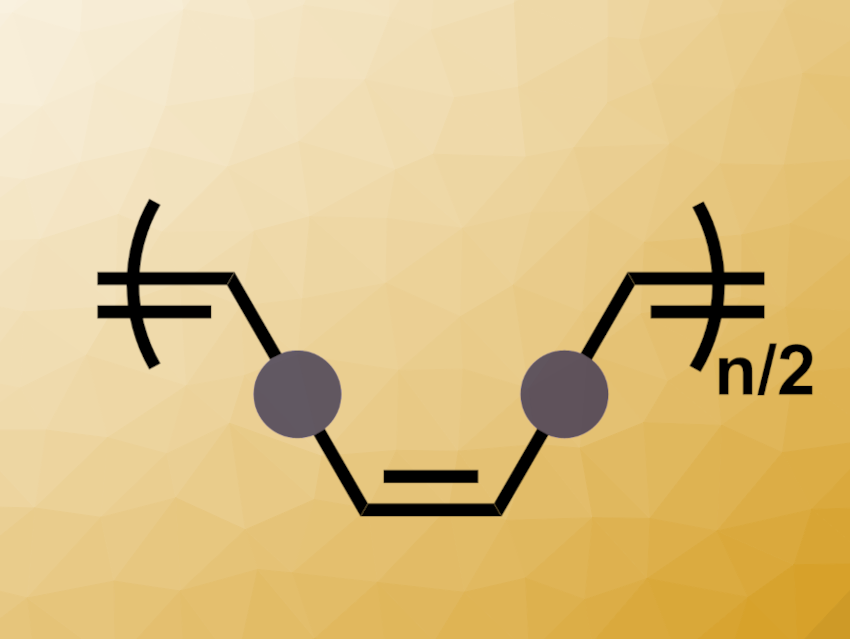
In olefin-containing polymers, the cis/trans stereochemistry can have effects on the mechanical and thermal properties of the materials. However, controlling this stereochemistry can be challenging. Existing approaches can suffer from drawbacks such as a limited functional group tolerance and a difficulty in achieving high cis selectivities. Methods based on olefin metathesis such as acyclic diene metathesis (ADMET) can be useful for the synthesis of olefin-containing polymers. However, ADMET typically produces polymers with mostly trans alkenes.
Quentin Michaudel, Texas A&M University, College Station, USA, and colleagues have developed a cis-selective ADMET polymerization of readily available α,ω-diene monomers with high functional group tolerance. The team used a cyclometalated ruthenium-based metathesis catalyst that contains an N-heterocyclic carbene with a 2,6-diisopropylphenyl substituent and an adamantyl group. At a reaction temperature of 23 °C in 1,2,4-trichlorobenzene (TCB), the team achieved a cis content of up to 99 %.
The reaction was used to prepare polycarbonates, polysulfites, polyethers, polyesters, a polyoctenamer, polysiloxanes, halogenated polymers, and polymers functionalized with alcohol groups. With this approach, the team prepared a range of cis-rich polyalkenamers and compared their properties with the corresponding trans-rich polymers. They found that a high cis content correlates with, e.g., increased thermal stability. Overall, the work could be useful for tuning the properties of olefin-containing polymers by controlling the cis/trans stereochemistry.
- cis-Selective Acyclic Diene Metathesis Polymerization of α,ω-Dienes,
Samuel J. Kempel, Ting-Wei Hsu, Jake L. Nicholson, Quentin Michaudel,
J. Am. Chem. Soc. 2023.
https://doi.org/10.1021/jacs.3c03978



I am interested in this article and kindly request a copy.
Dear Reader,
Thank you for your comment. You can find a link to the research article (where you can also find contact information for the authors) below the news text.
Your ChemistryViews Team